20. Inter-Related Effects
20.1. Introduction
20.1. Introduction
- This chapter of the Array Environmental Impact Assessment (EIA) Report presents the assessment of the likely significant effects (LSE1) (as per the “EIA Regulations”) on the environment in relation to inter-related effects as a result of the Ossian Array which is the subject of this application (hereafter referred to as “the Array”). Specifically, this chapter assesses the inter-related effects during the construction, operation and maintenance, and decommissioning phases.
- The assessments presented within this chapter have been informed by the individual chapters relevant assessment of effects and conclusions and their associated appendices in this Array EIA Report including:
- volume 2, chapter 7: Physical Processes;
- volume 2, chapter 8: Benthic Subtidal Ecology;
- volume 2, chapter 9: Fish and Shellfish Ecology;
- volume 2, chapter 10: Marine Mammals;
- volume 2, chapter 11: Offshore Ornithology;
- volume 2, chapter 12: Commercial Fisheries;
- volume 2, chapter 13: Shipping and Navigation;
- volume 2, chapter 14: Aviation, Military and Communications;
- volume 2, chapter 15: Infrastructure and Other Users;
- volume 2, chapter 18: Socio-Economics; and
- volume 2, chapter 19: Marine Archaeology.
20.2. Purpose of the Chapter
20.2. Purpose of the Chapter
- The Array EIA Report provides the Scottish Ministers, statutory and non-statutory stakeholders with adequate information to determine the LSE1 of the Array on the receiving environment. This is further outlined in volume 1, chapter 1.
- The purpose of this Inter-Related Effects Array EIA Report chapter is to describe:
- the receptor groups considered within the inter-related effects assessment;
- the potential for effects on receptor groups across the three key phases of the Array (construction, operation and maintenance and decommissioning);
- the potential for multiple effects on a receptor group, as presented within the topic specific chapter, to interact to create inter-related effects; and
- the inter-related effects across different trophic levels of the ecosystem, affecting the environment.
- This chapter follows the ecosystem-based approach, which is defined as “a strategy for the integrated management of land, water and living resources that promotes conservation and sustainable use in an equitable way” (Convention on Biological Diversity (CBD), 2023). The purpose of the ecosystem-based approach is to assess how the Array may interact at the ecosystem level, affecting the environment. This has been carried out following stakeholder advice received during the Scoping process (MD-LOT, 2023)
20.3. Study Area
20.3. Study Area
- Due to the differing spatial extent of effects experienced by different offshore receptors, the study area for potential inter-related effects varies according to topic and receptor. The likely significant inter-related effects assessed in Part One of this chapter are, therefore, also limited to the study area defined in each of the topic specific chapters outlined in section 20.1.
- As the largest study area relates to offshore ornithology, this is the maximum limit of the inter-related effects study area.
20.4. Policy and Legislative Context
20.4. Policy and Legislative Context
- Volume 1, chapter 2 of the Array EIA Report presents the policy and legislation of relevance to renewable energy infrastructure.
- Of relevance to the inter-related effects chapter, Article 3(1) for the EIA Directive requires that the interaction between the environmental factors (e.g. human health, biodiversity, land, soil, water, air and climate etc) must be identified, described and assessed in the Array EIA Report. Under the EIA Regulations, there is a requirement to consider inter-relationships between topics that may lead to environmental effects. Other than this, there is no policy relevant to inter-related effects in Scotland, thus this chapter has been compiled following advice from stakeholders as detailed in Table 20.1 Open ▸ .
20.5. Consultation
20.5. Consultation
- Table 20.1 Open ▸ presents a summary of the key issues raised during consultation activities undertaken to date specific to inter-related effects for the Array and in the Ossian Array Scoping Opinion (Marine Directorate – Licensing Operations Team (MD-LOT), 2023) along with how these have these have been considered in the development of this Array EIA Report chapter. Further detail is presented within volume 1, chapter 5.
Table 20.1: Summary of Issues Raised During Consultation and Scoping Opinion Representations Relevant to Inter-Related Effects
20.6. Data Sources
20.6. Data Sources
- The baseline environments for the receptor groups considered in section 20.8 of this chapter are specific to each receptor group and are, therefore, set out in the detail in the relevant topic chapters (see section 20.1).
- This chapter draws on the conclusions made within the technical chapters for the assessment of impacts acting in isolation on the receptor groups. The relevant sections drawn upon in this inter-related effects assessment are presented in the Array EIA Report in volume 2, chapters 7 to 19 (excluding volume 2, chapter 16).
- It should be noted that the inter-related effects impact assessment assesses the LSE1 from the Array only as set out in volume 2, chapters 7 to 20, and not those from other projects other than the Proposed offshore export cable. Inter-related effects from other projects are considered in the cumulative effects sections of the relevant chapters (volume 2, chapters 7 to 20).
- Specific guidance relevant to the approach to the assessment in each part of this chapter has been stated in section 20.7.
20.7. Assessment Methodology
20.7. Assessment Methodology
- NatureScot highlighted that “increasingly there is a need to understand potential impacts holistically at a wider ecosystem scale in addition to the standard set of discrete individual receptor assessments. ( Table 20.1 Open ▸ ) In response, the assessment presented in this chapter has been split into two parts, Part One: Receptor Based Inter-Related Effects Assessment outlines the assessment required as part of the EIA Regulations, and Part Two: Ecosystem Effects Assessment looks to address the request from NatureScot for a wider ecosystem scale assessment.
20.7.1. Part One: Inter-Related Effects Assessment Method
20.7.1. Part One: Inter-Related Effects Assessment Method
- The following sections present the approach used for the inter-related effects assessment of the Array. For the purpose of this assessment, the following definition has been used for the term inter-related effect:
- an “Inter-Related Effect” is when there are multiple effects upon the receptor arising from the Array, occurring either where a single effect acts upon a receptor over time to produce a potential additive effect or where a number of separate effects, such as underwater noise and habitat loss, affect a single receptor, for example marine mammals.
- Table 20.2 Open ▸ presents full definitions of the terms used in this assessment.
Table 20.2: Definitions of Array Lifetime and Receptor-Led Inter-Related Effects
Approach to assessment
- The following guidance documents have been followed relating to the assessment of inter-related effects:
- The Guidelines for the Assessment of Indirect and Cumulative Impacts as well as Impact Interactions (EC, 1999); and
- Institute of Environmental Management and Assessment (IEMA) Environmental Impact Assessment Guide to Shaping Quality Development (IEMA, 2016).
- The approach for assessing the potential inter-related effects on each receptor or receptor group follows the steps summarised in Table 20.3 Open ▸ and outlined.
Table 20.3: Staged Approach to Assessing Inter-Related Effects
Stage 1: Topic specific assessment
- The first stage of the assessment of inter-related effects is presented in each of the Array EIA Report topic chapters and comprises the individual assessments of effects on receptors across the construction, operation and maintenance and decommissioning phases of the Array.
Stage 2: Identification of receptor
- The second stage involved a review of the assessments undertaken in the topic specific chapters to identify the ‘receptor groups’ requiring assessment within this chapter. The term ‘receptor group’ is used to highlight that, for the purposes of the assessment, the potentially sensitive receptors have been grouped together e.g. marine mammals. The use of a ‘receptor group’ approach has been agreed with stakeholders via the Ossian Array Scoping Opinion (MD-LOT, 2023). The receptor groups assessed can be broadly categorised as those relating to the physical environment, the biological environment and the human environment, as follows (see paragraph 2 for references to chapters):
- physical environment:
– physical processes.
- biological environment:
– benthic subtidal ecology;
– fish and shellfish ecology;
– marine mammals; and
– offshore ornithology.
- human environment
– commercial fisheries;
– shipping and navigation;
– aviation, military and communications;
– infrastructure and other users;
– socio-economics; and
– marine archaeology.
Stage 3: Identification of potential inter-related impacts on receptor groups
- Following the identification of receptor groups, the potential inter-related impacts on those receptor groups were identified via a review of the assessment sections for each relevant topic chapter. The judgement to which impacts may result in inter-related effects upon receptors associated with the Array was based on the professional judgement and experience of the project management team.
- It is important to recognise potential linkages between the topic-specific chapters, whereby effects and receptor groups assessed in each chapter have the potential for secondary effects on any number of other receptors. Examples include:
- volume 2, chapter 8 addresses effects on benthic habitats and species arising from changes to the physical environment (as described in volume 2, chapter 7);
- volume 2, chapters 8 and 9 addresses effects on fish and shellfish receptors arising from the impacts on benthic habitats;
- volume 2, chapters 10 and 11 assess the effects on marine mammal and seabird receptors (respectively) arising from potential changes in benthic habitats and the distribution of fish, which from their principal prey (as described in volume 2, chapter 9);
- volume 2, chapter 13 addresses effects on shipping and navigation receptors arising from potential impacts as a result of a combination of effects caused by marine mammal collision (as described in volume 2, chapter 10);
- volume 2, chapter 14 assesses the effects on aviation and radar receptors arising from potential impacts on restriction of access to infrastructure by both helicopters and vessels (as described in volume 2, chapter 15); and
- volume 2, chapter 15 asses the effects on infrastructure and other user receptors arising from potential impacts on aggregate resource as a result of potential increase in suspended sediment concentrations (SSCs) and deposition and effects on sediment transport pathways (as described in volume 2, chapter 7).
- Where such linked relationships arise these have been fully assessed within the individual topic chapters. This chapter on inter-related effects therefore summarises the consideration of these inter-related effects on linked receptors already set out in the topic specific chapters.
- It should be noted that this chapter does not consider that there are likely to be any cumulative receptor-led effects from offshore and onshore activities associated with the Array. This is primarily due to the uncertainty around the location of the offshore export cable and the location of landfall for the export cable in Lincolnshire (at a significant distance from the Array). The close proximity of the offshore export cable occurs where the cable exits the site and the cumulative interaction will be reduced as the cable moves further away from the Array. Furthermore, despite the uncertainty around the location of the offshore export cable and location of the landfall for export cable in Lincolnshire, with the designed in measures in place for the Array, the Applicant considers it to be unlikely that there will be any cumulative receptor-led effects from offshore and onshore activities associated with the Array. However, taking a precautionary approach, the Applicant has assessed the cumulative potential for inter-related effects with the Proposed offshore export cable in section 20.8.2. This will be further assessed in a separate EIA Report for the offshore and onshore export cable works, following the agreement of the landfall site, Proposed offshore export cable corridor(s) and Proposed onshore transmission infrastructure.
Stage 4: Assessment of inter-related effects on each receptor group
- Individual effects on each of the key receptor groups have been identified across the three Array phases (i.e. Array lifetime effects) as well as the interaction of multiple effects on a receptor (i.e. receptor-led effects), as defined in Table 20.2 Open ▸ .
- The significance of the individual effects is presented in the summary of effects, mitigation measures and monitoring tables for each receptor group within the relevant topic chapters (all conclusions for significance of effect defined in the topic chapters assume successful implementation of mitigation measures where appropriate). A descriptive assessment of the scope for these individual effects to interact to create a different or greater effect is then undertaken (section 20.8). This assessment incorporates qualitative and, where possible, quantitative assessments. Each topic-specific assessment, outlined in Table 20.4 Open ▸ to Table 20.14 Open ▸ , presents an assessment of the significance of effect to any such inter-related effect.
- The inter-related effects assessment presents and utilises the maximum significant adverse effects for the Array (i.e. the maximum design scenarios including successful implementation of measures adopted as part of the Array where appropriate), noting that individual effects may not be significant at the topic-specific level but could become significant when their inter-related effect is assessed. Effects of moderate significance or above are therefore considered important in the decision-making process, whilst effects of minor significance or less warrant little, if any, weight in the decision-making process. Effects of moderate significance or above may occur in only one phase of the lifetime of the Array (e.g. during the construction phase but not the operation and maintenance or decommissioning phases). Where this is the case, it has been made clear that, as a result, there will be no inter-related effects across the lifetime of the Array.
20.7.2. Part Two: Ecosystem Based Effects Assessment Method
20.7.2. Part Two: Ecosystem Based Effects Assessment Method
- The purpose of the ecosystem-based assessment is to qualitatively assess the potential effects of the Array at the ecosystem level, to better understand how predator – prey relationships could be altered and how this could impact the functioning of the ecosystem.
- This structure of Part Two: Ecosystem Effects Assessment is as follows:
- overview;
- ecosystem baseline;
- the marine food web;
- the key predator species;
- the key prey species;
- how the food system works;
- future ecosystem baseline;
- existing pressures on prey species;
- effects of the Array on prey species; and
- effects of the Array on predator species.
- Information and conclusions from the relevant chapters of the Array EIA Report and their corresponding technical reports have been used to build up a picture of the marine ecosystem in the locality of the Array and inform the baseline for the ecosystem assessment. This information has also been used to inform the assessments within these sections to ultimately conclude whether the Array, and cumulatively with other plans and projects, is likely to result in changes to prey species which in turn will result in changes to predator species and therefore result in likely significant ecosystem effects.
- As new surveys and studies bridge knowledge gaps, we are developing a greater understanding of how offshore wind farms impact marine ecosystems. Details of two research programmes are provided below, the outcomes of which are and will be used to inform policy measures and provide guidance and recommendations on improving stakeholder confidence in the sustainable delivery of offshore wind farms. Furthermore, these projects are key to informing ecosystem assessment approach.
PrePARED Project
- Following guidance from the Scottish Ministers, detailed in Table 20.1 Open ▸ , the PrePARED project (PrePARED, 2022) has been used to assist in the understanding of predator-prey relationships in and around fixed offshore wind farms.
- Funded by the Offshore Wind Evidence and Change Programme and Crown Estate Scotland, the PrePARED project seeks to better understand how seabirds and marine mammals respond to offshore wind developments and the mechanisms underpinning their responses, particularly changes in prey distribution. An increased certainty on the magnitude of cumulative impacts will facilitate deployment of offshore wind farms at the pace and scale required to help meet the United Kingdom (UK) government’s renewable energy targets and reach net-zero emissions.
- At the time of writing this chapter, the 2022 Annual Report was the latest version available from PrePARED which summarises the first year of the five-year project (PrePARED, 2022). Part of the project activities included fish, seabird and marine mammal data collection in and around offshore wind farms in the Firth of Forth and Tay and the Moray Firth. Examples of tasks conducted as part of the project include:
- surveying fine-scale fish response to offshore wind farms;
- collating historical data to determine large-scale fish distribution;
- collating historical data to develop seabird spatial distribution models;
- designing acoustic telemetry arrays to track fish and marine mammal movement; and
- collating historical data from various methods to develop marine mammal distribution models.
Ecological Consequences of Offshore Wind (ECOWind)
- The ECOWind research programme seeks to investigate all possible effects of offshore wind farms on marine biodiversity (ECOWind, 2024). ECOWind are currently undertaking four projects:
- BOWIE
– The BOWIE project will use a combination of environmental and social research to gain knowledge on the impact of wind expansion on seabed invertebrate and fish species, taking into consideration other pressures on the marine environment including construction noise and vibration, electromagnetic fields and elevated temperatures associated with cabling.
- ECOWind ACCELERATE
– The ECOWind ACCELERATE project will investigate the ecological implications of accelerated seabed mobility around windfarms. Scientists will be studying the combined impacts of climate change and offshore wind farms on seabed habitats and organisms and how baseline changes can cause affect the wider marine ecosystem around the UK.
- ECOWINGS
– The ECOWINGS project will investigate the cumulative effects of offshore wind on key seabird species, establishing pathways for strategic compensation to ensure net gain for seabird populations and the wider marine ecosystem.
- PELAgIO
– The PELAgIO project will investigate the impacts that offshore wind development can have on the marine food chain.
20.8. Part One: Receptor Based Inter-Related Effects Assessment
20.8. Part One: Receptor Based Inter-Related Effects Assessment
20.8.1. Assessment of Inter-Related Effects
20.8.1. Assessment of Inter-Related Effects
- For each of the receptor groups listed above, the scope for impact to these receptors to create Array lifetime effects over all phases and/or receptor-led effects through interacting together on a particular group has been explored and discussed in the following sections.
- The sections have been subdivided into the categories listed in paragraph 21.
Physical environment
Physical processes
- For physical processes, the following potential impacts have been considered within the inter-related effects assessment:
- increase in SSCs and associated deposition and sediment transport due to operation and maintenance activities;
- impacts to the wind field due to the presence of infrastructure; and
- impacts to seasonal stratification due to the presence of infrastructure.
- Table 20.4 Open ▸ lists the inter-related effects (Array lifetime effects) that are predicted to arise during the construction, operation and maintenance and decommissioning phase of the Array and also the inter-related effects (receptor-led effects) that are predicted to arise for physical processes receptors.
- Physical processes receptors have the potential to have secondary effects on other receptors and these effects are fully considered in the topic specific chapters and elsewhere in this chapter. These receptors and effects are:
- benthic subtidal ecology:
– increased SSCs; and
– increased sediment deposition; and
– changes to seasonal stratification.
- fish and shellfish ecology:
– increased SSCs;
– increased sediment deposition; and
– changes to seasonal sediment deposition.
- marine mammals:
– increased SSCs;
– sediment deposition; and
– changes to seasonal stratification.
- infrastructure and other users:
– increased SSCs.
Table 20.4: Summary of Likely Significant Potential Inter-Related Effects for Physical Processes from Individual Effects Occurring across the Construction, Operation and Maintenance and Decommissioning Phases of the Array (Array Lifetime Effects) and from Multiple Effects Interacting Across all Phases (Receptor-led Effects)
Biological environment
Benthic subtidal ecology
- For benthic subtidal ecology, the following potential impacts have been considered within the inter-related effects assessment:
- temporary habitat loss and disturbance;
- long term habitat loss and disturbance;
- increased SSCs and associated deposition;
- effects to benthic subtidal ecology from electromagnetic field (EMF) from subsea electrical cabling;
- colonisation of hard substrates;
- effects to benthic subtidal ecology due to removal of hard substrates; and
- increased risk of invasive non-native species (INNS).
- Table 20.5 Open ▸ lists the inter-related effects (Array lifetime effects) that are predicted to arise during the construction, operation and maintenance and decommissioning phase of the Array and also the inter-related effects (receptor-led effects) that are predicted to arise for benthic subtidal ecology receptors.
- Benthic subtidal ecology receptors have the potential to have secondary effects on other receptors and these effects are fully considered in the topic specific chapters and elsewhere in this chapter. These receptors and effects are:
- fish and shellfish ecology;
– temporary (during construction, operation and maintenance and decommissioning phases), long term (during operation and maintenance phase only) and permanent habitat alteration (post decommissioning) habitat loss and disturbance;
- marine mammals;
– effects on marine mammals due to altered prey availability; and
- offshore ornithology;
– – changes to prey availability.
Table 20.5 Summary of Likely Significant Potential Inter-Related Effects for Benthic Subtidal Ecology from Individual Effects Occurring across the Construction, Operation and Maintenance and Decommissioning Phases of the Array (Array Lifetime Effects) and from Multiple Effects Interacting Across all Phases (Receptor-led Effects)
Fish and shellfish ecology
- For fish and shellfish ecology, the following potential impacts have been considered within the inter-related effects assessment:
- temporary habitat loss and disturbance;
- long-term habitat loss and disturbance;
- effects to fish and shellfish receptors due to EMF from subsea electrical cabling;
- colonisation of hard structures;
- underwater noise impacting fish and shellfish receptors; and
- underwater noise from the operation of floating wind turbines and anchor mooring lines impacting fish and shellfish receptors.
- Table 20.6 Open ▸ lists the inter-related effects (Array lifetime effects) that are predicted to arise during the construction, operation and maintenance and decommissioning phase of the Array and also the inter-related effects (receptor-led effects) that are predicted to arise for fish and shellfish ecology receptors.
- Fish and shellfish ecology also have the potential to have a secondary effect on other receptors and these effects are fully considered in the topic-specific chapters and elsewhere in this chapter. These receptors and effects are:
- marine mammals;
– changes in the fish and shellfish community resulting from the impacts during construction, operation and maintenance, and decommissioning may lead to the loss of prey resources for marine mammals resulting in effects of negligible significance, which are not significant in EIA terms (volume 2, chapter 10);
- offshore ornithology;
– changes in the fish and shellfish community resulting from the impacts during construction, operation and maintenance, and decommissioning may lead to the changes in prey availability for birds. However, as noted in volume 2, chapter 11, there are negligible to minor effects on fish and shellfish ecology and therefore negligible or minor effects are predicted for offshore ornithology, which are not significant in EIA terms.
- commercial fisheries;
– changes in fish and shellfish community resulting from impacts during construction, operation and maintenance, and decommissioning may affect commercial fisheries receptors by effects on access to target species, however as noted in this chapter, there are negligible or minor effects on fish and shellfish receptors therefore negligible or minor effects are predicted for commercial fisheries, which are not significant in EIA terms.
Table 20.6: Summary of Likely Significant Inter-Related Effects for Fish and Shellfish Ecology from Individual Effects Occurring Across the Site Preparation and Construction, Operation and Maintenance and Decommissioning Phases of the Array (Array Lifetime Effects) and from Multiple Effects Interacting Across all Phases (Receptor-led Effects)
Marine mammals
- For marine mammals, the following potential impacts have been considered within the inter-related effects assessment:
- injury and disturbance from underwater noise generated during piling;
- injury and disturbance from underwater noise generated during UXO clearance;
- injury and disturbance due to site investigation surveys (including geophysical surveys);
- Injury and disturbance to marine mammals from elevated underwater noise due to vessel use and other activities.
- injury due to collision with vessels;
- effects on marine mammals due to EMFs from subsea electrical cabling in the water column;
- injury and disturbance from underwater noise generated during the operation of floating wind turbines and anchor mooring lines;
- effects on marine mammals due to entanglement associated with the Array; and
- effects on marine mammals due to altered prey availability.
- Table 20.7 Open ▸ lists the inter-related effects (Array lifetime effects) that are predicted to arise during the construction, operation and maintenance, and decommissioning phases of the Array and also the inter-related effects (receptor-led effects) that are predicted to arise for marine mammal receptors.
Table 20.7: Summary of Likely Significant Inter-Related Effects for Marine Mammals from Individual Effects Occurring Across the Site Preparation and Construction, Operation and Maintenance and Decommissioning Phases of the Array (Array Lifetime Effects) and from Multiple Effects Interacting Across all Phases (Receptor-led Effects)
Stressor 1: injury or disturbance from elevated underwater noise (from piling, UXO clearance, site-investigation surveys, vessels, operational noise from turbines/mooring lines)
- During the construction phase activities resulting in elevated underwater noise include piling, UXO clearance, site investigation surveys and vessel movements could occur. These activities are likely to result in disturbance to marine mammals which may be additive in nature if activities are synchronised, as it could lead to a larger area disturbed at any one time. Disturbance is likely to occur as short term, localised events for each activity within the construction phase. Prior to piling, for example, UXO clearance could result in no more than 15 single clearance events, and disturbance occurring mainly during secondary mitigation (ADDs and soft start) rather than the UXO clearance event itself which would be no more than seconds for each. There is also a small potential that animals could experience injury during UXO clearance (due to an accidental high order detonation). Site investigation surveys are likely to occur over a total duration of up to five months (over a three year period) whilst disturbance during vessel activity will occur intermittently throughout this phase with timings linked to the pre-construction activities (UXO and site-investigation surveys).
- During the construction phase, activities resulting in elevated underwater noise include piling, other construction activities and vessel movements could occur. Since injury to marine mammals will be mitigated through the Marine Mammal Mitigation Protocol (MMMP) (volume 4, appendix 22), the key focus is on disturbance effects. Disturbance could occur intermittently on a total of 602 days over the construction phase of 96 months. Other construction activities (e.g. drilling and cable laying) and vessel movements would occur intermittently within the eight year construction phase. When piling occurs the disturbance effects are likely to be greater than for any of the other activities contributing to elevated underwater noise so there is less likely to be an additive or synergistic effect during piling. There may, however, be an additive effect spatially where two or more noise-producing activities occur in different parts of the Array, or temporally due to ongoing disturbance from activities throughout the construction phase (e.g. if they occur consecutively).
- During the operation and maintenance phase, activities resulting in elevated underwater noise include vessel activity, geophysical surveys and operational noise from floating turbines and mooring lines. These activities have the potential to result in disturbance to marine mammals which may be additive if activities are synchronised, as it could lead to a larger area disturbed at any one time. Disturbance is likely to occur as short term, localised events for vessel activity and geophysical surveys and the disturbance from operational noise is expected to be minimal, but there may be an additive effect spatially where two or more noise-producing activities occur in different parts of the Array, or temporally due to ongoing disturbance from activities throughout the operation and maintenance phase (e.g. if they occur consecutively).
- During decommissioning, vessel movements associated with decommissioning activities will result in elevated underwater noise which could lead to disturbance to marine mammals. Disturbance is likely to occur as short term, localised events and there may be an additive effect spatially where vessels are operating in different parts of the Array, or temporally due to ongoing disturbance throughout the decommissioning phase.
- Therefore, marine mammal receptors have the potential to experience ongoing disturbance due to elevations in underwater noise from different sources at all phases of the Array. The sensitivity of key species will be linked to their ability to tolerate the stressor such that their ability to function normally (e.g. forage, reproduce, communicate, avoid predators) is not impeded. The assessment, which adopts a highly precautionary approach, demonstrated that for all impacts, considered in isolation, the residual effects will not be significant (after implementation of secondary mitigation) as either the spatial scale is very localised or where larger scale effects do occur (i.e. during piling or UXO) these will be highly reversible with animals returning to baseline levels rapidly. After implementation of secondary mitigation there is, however, potentially a small residual number of harbour porpoise Phocoena phocoena that could experience auditory injury during UXO clearance activities and would represent only a very small proportion of the NS MU population.
- There are, however, uncertainties as to how all activities interact to contribute to an additive effect from underwater noise as a stressor. In a Before-After-Control-Impact design (BACI) study looking at foraging activity of harbour porpoise between baseline periods and different construction phases of the Beatrice and Moray East Offshore Wind Farms (Benhemma-Le Gall et al., 2021) an eight to 17% decline in harbour porpoise occurrence in the impacted area during pile-driving and other construction activities was observed, with probability of detection negatively related to levels of vessel intensity and background noise.
- To some extent it is anticipated that animals will acclimatise to or compensate for such increases in underwater noise. Graham et al. (2019), for example, demonstrated acclimatisation in harbour porpoise. The study showed that the proportional response of harbour porpoise to piling noise decreased over the piling phase, with the proportion of animals disturbed at a received level of 160 dB re 1 µPa decreased from 91.5% to 49.2% from the first pile to the last pile. Kastelein et al. (2019) suggest that harbour porpoise (a species with high daily energy requirements) may be able to compensate for period of disturbance as they can dramatically increase their food intake in a period following fasting within out any detriment to their health. In the Moray Firth, harbour porpoises displaced during wind farm construction of Beatrice and Moray East Offshore Wind Farms increased their buzzing activity, potentially compensating for lost foraging opportunities (although there may be an additional energetic cost from the fleeing and distance travelled to compensate for) (Benhemma-Le Gall et al., 2021).
- Therefore, as detailed in paragraphs 50 to 56 above, significance is considered to be minor adverse and therefore not significant in EIA terms.
Stressor 2: injury due to collisions with vessels
- Injury due to collisions with vessels is associated with increased vessel movement, the impact of which was assessed from different types of vessels and at different phases of the Array. As described in paragraph 50 et seq., over the lifetime of the Array there will be a longer term risk to marine mammal receptors however, with designed in measures in place the potential of experiencing injury is likely to be reduced and therefore it is not anticipated that an additive effect will occur. Additionally, to some extent the noise from the vessels themselves (Stressor 2, paragraph 50 et seq.) would act antagonistically with this impact by deterring animals away from vessels and thereby further reducing the risk of injury due to collision. Furthermore, marine mammals in this area are already accustomed to high level of vessel activity. For example, Buckstaff (2004) demonstrated that bottlenose dolphins Tursiops truncastus increased their rate of whistle production at the onset of a vessel approach, and then decreased production during and after it had passed. This increased whistle production may be a tactic to reduce signal degradation to ensure that information is being communicated in elevated noisy environment, but it also demonstrates that animals are aware of approaching vessel from a distance. This corroborates previous research of Nowacek et al. (2001) found that bottlenose dolphins swim in tighter aggregated groups during vessel approaches, therefore if a vessel is loud enough to be detected by an animal for which it adjusts its behaviour, the likelihood of collision decreases.
- Therefore, as detailed in paragraph 58, significance is considered to be minor adverse and therefore not significant in EIA terms.
Stressor 3: EMF
- EMF is highly localised and there is limited information on the effect of EMF on marine mammal receptors. It is unlikely to be additive with other stressors, given it will be confined to very specific locations in close proximity to the cables. There may be some synergistic effects if animals moving away from other disturbance activities (such as vessels) dive down and therefore move closer to the inter-array cables. Therefore, significance is considered to be minor adverse and therefore not significant in EIA terms.
Stressor 4: entanglement
- The risk of entanglement is highly localised. The possibility of primary entanglement is very unlikely given design factors such as the taut mooring lines with high bending stiffness (Statoil, 2015) and low weight of the cable systems (Synthesis of Environmental Effects Research (SEER), 2022). It is noted there is limited information to assess entanglement of marine mammal receptors in offshore wind development to date. Injury from entanglement is very different to other types of injury (e.g. injuries from collision, PTS) and therefore there is not considered to be any additive effects. As is the case for stressor 2, to some extent the noise (pinging or snapping) from operational noise from turbines/mooring lines and any vessels utilised during the operation and maintenance phase themselves may act antagonistically with this impact by deterring animals away from the mooring lines. Therefore, significance is considered to be minor adverse and therefore not significant in EIA terms.
Stressor 5: changes in prey communities.
- The EIA considered overall effect on fish and shellfish communities from multiple stressors (i.e. habitat loss, SSC, underwater noise, EMF etc) (see volume 3, chapter 9) and therefore, in this respect, has taken an ecosystem-based approach. For some, stressors such as underwater noise effects on fish and shellfish, will be over the same timescales as marine mammals whilst for others, such as temporary habitat loss, timescales may be different to those assessed for marine mammals (e.g. low mobility or sessile species may recover slowly). The assessment of effects, however, demonstrated that due to the high mobility of marine mammals, generalist feeding strategy and ability to exploit different prey species, combined with the small scale of potential changes in context of wider available habitat, the changes to fish and shellfish communities are unlikely to have an effect even from multiple stressors. Therefore, significance is considered to be minor adverse and therefore not significant in EIA terms.
Multiple stressors: inter-related effect of all stressors
- Arrigo et al. (2020) studied synergistic interactions among growing stressors to an Arctic ecosystem and found that synergistic interactions amplify adverse stressor effects, and the impact of synergy is predicted to increase with the magnitude of stressors. Arrigo et al. (2020) suggests that large organisms at higher trophic levels, such as marine mammals, tend to be generally negatively impacted by increasing stressor interaction strength but the variability in the response to stressor is small and therefore reduces the probability of population collapse.
- For stressor 1 (elevated underwater noise), there is the potential for marine mammals to forage in different habitats and to compensate for reduced foraging time. As such the ability of displaced animals will depend on the availability of prey resources in the habitat to which the animals are displaced. Studies have shown that for small, localised marine mammal populations with high site fidelity, there may be biological risks posed by displacement (Forney et al., 2017). For example, due to the importance of the areas for survival (i.e. areas of high resource availability), animals may be highly motivated to remain in an area despite adverse impacts which may increase stress (Rolland et al., 2012). Thus, the inter-related effects of underwater noise and changes in fish and shellfish prey resources needs to be considered. Impacts on fish and shellfish prey resources (stressor 5) were predicted to be localised and short-term and therefore unlikely to contribute to an inter-related effect where animals are displaced beyond the site boundary. Within the site boundary however, there may be short term inter-related effects of noise disturbance and reduced fish and shellfish prey resources. For marine mammals remaining in proximity to the Array, a substantial disruption in foraging may not be easy to compensate for where there are shifts in the species composition or localised reductions of fish and shellfish communities. It has been suggested it may be possible that damaged or disoriented prey could attract marine mammals to an area of impact due to providing short term feeding opportunities but increasing levels of exposure (Gordon et al., 2003) however, there is currently little evidence available to investigate such indirect effects on marine mammals.
- The assessment has largely described potential adverse effects but there is also potential for some beneficial effects on marine mammal receptors. Construction of offshore wind farms can lead to the introduction of hard substrates which can lead to the establishment of new species and new fauna communities, and this may in turn attract marine mammals (Fowler et al., 2018, Lindeboom et al., 2011, Raoux et al., 2017). Consequently, even where there is potential for an inter-related effect between ongoing vessel noise during the operation and maintenance phase this may be compensated for, to some extent, by an increase in available prey resources. Russell et al. (2014) and Russell and McConnell (2014) demonstrated that harbour seals Phoca vitulina and grey seals Halichoerus grypus moved between hard structures at two operational wind farms and used space-state models to predict where animals were remaining at these locations to actively forage and where they were travelling to the next foundation structure. Lindeboom et al. (2011) studied the ecological effects of the Egmond aan Zee Offshore Wind Farm and found that even though the fish community was highly dynamic in time and space, with only minor effects upon fish assemblages observed during the operation and maintenance phase, some fish species (e.g. cod Gadus morhua) benefited from the ‘shelter’ within the wind farm, although this effect may be reduced for floating wind turbines. This is likely due to reduced fishing activity and the new hard substratum with associated fauna which attracts predator species. Lindeboom et al. (2011) suggested the observed increase in echolocation activity of harbour porpoise within the wind farm may be correlated with presence of additional increased food sources compared to reference areas (Lindeboom et al., 2011).
- The potential inter-related effects between underwater noise and collision risk have been discussed previously (in paragraph 58) and it is considered likely that marine mammals will move away from moving vessels in response to engine noise, therefore reducing the risk of collision (classed as an antagonistic interaction). Alternatively, marine mammals may tolerate and persist in a highly stressed state (as a result of injury caused by underwater noise) while the vessels are approaching (Muto et al., 2018). Animals could also become habituated to vessel noise and not move away from the vessel (McWhinnie et al., 2018) which would result in a synergistic interaction (Weilgart, 2011). Therefore, the outcome will depend on the degree of habituation and prior-experience and a number of acoustical properties that allow an approaching vessel to be detected by a marine mammal species (Gerstein et al., 2005). However, as described in the impact assessment, with measures adopted as part of the Array (e.g. the VMP) in place it is likely that any risk of injury from collision with vessels will be negligible.
- Evidence for the potential long-term effects of offshore wind farms on marine mammals (related to all potential stressors) comes from monitoring programmes which baseline levels of abundance to construction and post-construction (operation and maintenance) phases. Few monitoring studies regarding impacts on marine mammals have been carried out to date.
- Aerial survey haul-out counts were conducted before, during and after the construction phases at Scroby Sands Offshore Wind Farm, off the coast of Norfolk, to monitor harbour and grey seal counts at haul-out site, located less than two kilometres away from the offshore wind farm array (Skeate et al., 2012). The two studies reported a decline in harbour seal numbers during construction, with numbers remaining lower over several subsequent years. However, the numbers of grey seals increased dramatically year after year throughout the construction and early operational periods. It has been suggested that it is possible that changes in harbour seal numbers may be linked to rapid colonisation of competing grey seal (Skeate et al., 2012). It was noted regional changes in patterns of haul-outs of harbour seal in the Wash coincided with the construction of the Scroby Sands Offshore Wind Farm, but such changes in harbour seal number could have been part of wider regional dynamics (Verfuss et al., 2016). It should be noted that Scroby Sands Wind Farm is located 2.5 km off the coast of Great Yarmouth whereas the Array is located 80 km offshore and therefore a greater distance from haul-out sites. As a part of marine mammal monitoring at Robin Rigg Offshore Wind Farm, boat-based surveys for cetaceans were conducted before, during, and after construction (Canning et al., 2013). The monitoring data suggested that harbour porpoise were displaced from the wind farm site during the construction phase and operation period when compared to the pre-construction numbers. However, because there was only one year of pre-construction survey, natural variation cannot be ruled out as the reason for the observed change, especially since control survey locations outside of the wind farm also appeared to experience declines in harbour porpoise density.
- With the rapid expansion of offshore wind farms, post-construction monitoring programmes are being implemented at various developments in Europe. Tougaard et al. (2003) studied short-term effects of the construction of wind turbines on harbour porpoises at Horns Rev Offshore Wind Farm. The study showed a decrease in porpoise acoustic activity within the wind farm at the onset of piling operations and subsequent recovery to higher levels a few hours after each piling operation was completed (Tougaard et al., 2003). (Tougaard et al., 2003) also showed that over the entire construction phase at Horns Reef there was no significant change in the abundance of harbour porpoise in the wind farm area compared to reference areas. Teilmann et al. (2008) also reported that during the operation and maintenance phase porpoise activity was higher in both the wind farm and reference area compared to baseline levels. As a result of monitoring at Nysted Offshore Wind Farm, it was demonstrated initially during construction and the first two years of operation that there were lower acoustic detections of harbour porpoises in the wind farm area, with recovery starting to occur within two years after the end of construction (Teilmann et al., 2006). Teilmann et al. (2006) suggested that animals were gradually habituating and returning to the wind farm area.(Teilmann et al., 2006).
- Nabe-Nielsen et al. (2011) suggested, using simulations of the response of harbour porpoise to wind farm construction, that wind farms already existing off Danish coast do not have impact on harbour porpoise population dynamics and that the that construction of new wind farms is not expected to cause any changes in the long-term dynamics of the population. Likewise, Edrén et al. (2010) and McConnell et al. (2012) investigated possible interactions between seals and Danish offshore wind farms (Nysted Wind Farm and Rødsand II) and found that although there was a temporary reduction in the number of seals hauled out during construction operations (i.e. piling), there was no long-term effect on haul-out behaviour trends.(Edrén et al., 2010)
- The examples of monitoring studies given in paragraphs 69 and 70 suggest marine mammal receptors can quickly recover and return to the impacted area, despite the potential effects from multiple stressors associated with offshore wind farms. Therefore, as detailed in paragraphs 63 to 70, significance is considered to be minor adverse and therefore not significant in EIA terms.
Offshore ornithology
- For offshore ornithology, the following potential impacts have been considered within the inter-related effects assessment:
- temporary habitat loss and disturbance;
- indirect habitats from construction/decommissioning noise;
- indirect impacts from UXO clearance;
- disturbance and displacement from the physical presence of wind turbines and maintenance activities;
- barrier to movement;
- collision with wind turbines;
- changes to prey availability; and
- entanglement.
- Table 20.8 Open ▸ lists the inter-related effects (Array lifetime effects) that are predicted to arise during the construction, operation and maintenance, and decommissioning phases of the Array and also the inter-related effects (receptor-led effects) that are predicted to arise for offshore ornithology receptors.
- Effects on offshore ornithology receptors are not expected to have secondary effects on other receptors.
Table 20.8: Summary of Likely Significant Inter-Related Effects for Offshore Ornithology from Individual Effects Occurring Across the Site Preparation and Construction, Operation and Maintenance and Decommissioning Phases of the Array (Array Lifetime Effects) and from Multiple Effects Interacting Across all Phases (Receptor-led Effects)
Human environment
Commercial fisheries
- For commercial fisheries, the following potential impacts have been considered within the inter-related effects assessment:
- temporary loss or restricted access to fishing grounds;
- long term loss or restricted access to fishing grounds;
- displacement of fishing activity into other areas;
- interference with fishing activity;
- increased snagging risk, which could result in loss or damage to fishing gear;
- increased steaming/vessel transit times; and
- impacts to commercial exploited species populations.
- Table 20.9 Open ▸ lists the inter-related effects (Array lifetime effects) that are predicted to arise during the construction, operation and maintenance, and decommissioning phases of the Array and also the inter-related effects (receptor-led effects) that are predicted to arise for commercial fisheries receptors.
- Effects on commercial fishing also have the potential to have a secondary effect on other receptors and these effects are fully considered in the topic-specific chapters and elsewhere in this chapter. These receptors and effects are:
- fish and shellfish ecology:
– displacement of fishing activities into other areas could increase fishing pressure in these areas and affect fish and shellfish receptors; and
- benthic subtidal ecology:
– displacement of fishing activities into other areas could increase fishing pressure in these areas and affect benthic subtidal ecology receptors; and
- socio-economics:
– reduced value of fish caught by commercial fisheries with potential downstream impacts, for example on fish processors.
Table 20.9: Summary of Likely Significant Inter-Related Effects for Commercial Fisheries from Individual Effects Occurring Across the Site Preparation and Construction, Operation and Maintenance and Decommissioning Phases of the Array (Array Lifetime Effects) and from Multiple Effects Interacting Across all Phases (Receptor-led Effects)
Shipping and navigation
- increased vessel to vessel collision risk resulting from displacement (third party to third party);
- increased vessel to vessel collision risk resulting from displacement (third party to Array vessels);
- vessel to structure allision risk; and
- reduced access to local ports and harbours.
- Table 20.10 Open ▸ lists the inter-related effects (receptor-led effects) that are predicted to arise for shipping and navigation receptors. No inter-related effects (Array lifetime effects) are predicted to arise during the construction, operation and maintenance and decommissioning phase of the Array since the potential impacts listed above in paragraph 78 will not be further exacerbated over the lifetime of the Array.
- Shipping and navigation receptors also have the potential to have secondary effects on other receptors and these effects are fully considered in the topic-specific chapters and elsewhere in this chapter. These receptors and effects are:
- commercial fisheries
– displacement from fishing grounds for commercial fishing vessels due to the presence of the buoyed construction and decommissioning areas during the construction and decommissioning phases, respectively; and
– displacement from fishing grounds for commercial fishing vessels due to the floating wind turbines and anchor mooring lines during the operation and maintenance phase.
Table 20.10: Summary of Likely Significant Potential Inter-Related Effects for Shipping and Navigation from Individual Effects Occurring across the Construction, Operation and Maintenance and Decommissioning Phases of the Array (Array Lifetime Effects) and from Multiple Effects Interacting Across all Phases (Receptor-led Effects)
Aviation, military and communications
- For aviation, military and communications, the following potential impacts have been considered within the inter-related effects assessment:
- creation of a physical obstruction to aircraft operations (including airborne SAR and low flying aircraft); and
- wind turbines causing interference on aviation radar systems (including PSR and ADR).
- Table 20.11 Open ▸ lists the inter-related effects (Array lifetime effects) that are predicted to arise during the construction, operation and maintenance, and decommissioning phases of the Array and also the inter-related effects (receptor-led effects) that are predicted to arise for aviation, military and communications receptors.
- The Array alone impacts were assigned residual significance of impact of no greater than minor adverse once mitigation is applied. It is therefore anticipated that the significance of combined effects on airspace and radar users will not be of any greater significance than the effects when assessed in isolation.
Table 20.11: Summary of Likely Significant Potential Inter-Related Effects for Aviation, Military and Communications from Individual Effects Occurring across the Construction, Operation and Maintenance and Decommissioning Phases of the Array (Array Lifetime Effects) and from Multiple Effects Interacting Across all Phases (Receptor-led Effects)
Infrastructure and other users
- For infrastructure and other users, the following potential impacts have been considered within the inter-related effects assessment:
- physical restrictions on space for recreational craft/recreational fishing vessels; and
- physical impact or loss of access to existing cables and pipelines.
- Table 20.12 Open ▸ lists the inter-related effects (Array lifetime effects) that are predicted to arise during the construction, operation and maintenance, and decommissioning phases of the Array and also the inter-related effects (receptor-led effects) that are predicted to arise for infrastructure and other users.
- As previously mentioned in paragraph 23, infrastructure and other users receptors are linked to physical processes and aviation, military and communication receptors. The inter-related effects have been fully in assessed in volume 2, chapter 7 and volume 2, chapter 14 of this Array EIA Report, respectively.
- Effects on infrastructure and other users have the potential to lead to secondary effects on other receptors and these effects are fully considered in the topic specific chapters and elsewhere in this chapter. These receptors are as follows:
- shipping and navigation
– displacement of recreational sailing and motor cruising, recreational fishing (boat angling) and other recreational activities (diving vessels); and
– physical impacts or loss of access to existing cables and pipelines.
Table 20.12: Summary of Likely Significant Potential Inter-Related Effects for Infrastructure and Other Users from Individual Effects Occurring across the Construction, Operation and Maintenance and Decommissioning Phases of the Array (Array Lifetime Effects) and from Multiple Effects Interacting Across all Phases (Receptor-led Effects)
Socio-economics
- For socio-economics, the following potential impacts have been considered within the inter-related effects assessment:
- employment and GVA impacts associated with the construction, operation and maintenance and decommissioning of the Array; and
- demographic changes and demand for housing and other services.
- Table 20.13 Open ▸ lists the inter-related effects (Array lifetime effects) that are predicted to arise during the construction, operation and maintenance, and decommissioning phases of the Array and also the inter-related effects (receptor-led effects) that are predicted to arise for socio-economic receptors.
Table 20.13: Summary of Likely Significant Potential Inter-Related Effects for Socio-economics from Individual Effects Occurring across the Construction, Operation and Maintenance and Decommissioning Phases of the Array (Array Lifetime Effects) and from Multiple Effects Interacting Across all Phases (Receptor-led Effects)
Marine archaeology
- For marine archaeology, the following potential impacts have been considered within the inter-related effects assessment:
- sediment disturbance and deposition leading to indirect impacts on marine archaeology receptors; and
- alteration of sediment transport regimes.
- Direct damage to marine archaeology receptors has not been assessed as part of the inter-related effects assessment as there is no potential for direct damage to accumulate through the lifetime of the project or to interact, spatially and temporally, to create inter-related effects on a receptor.
- Table 20.14 Open ▸ lists the inter-related effects (Array lifetime effects) that are predicted to arise during the construction, operation and maintenance, and decommissioning phases of the Array and also the inter-related effects (receptor-led effects) that are predicted to arise for infrastructure and other users.
- Effects on marine archaeology do not have the potential to lead to secondary effects on other receptors.
Table 20.14: Summary of Likely Significant Potential Inter-Related Effects for Marine Archaeology from Individual Effects Occurring across the Construction, Operation and Maintenance and Decommissioning Phases of the Array (Array Lifetime Effects) and from Multiple Effects Interacting Across all Phases (Receptor-led Effects)
20.8.2. Cumulative Effects Assessment
20.8.2. Cumulative Effects Assessment
- As mentioned in paragraph 25, the inter-related effects from other projects are considered in the cumulative effects sections of the relevant chapters (volume 2, chapters 7 to 20). However, the cumulative effects of the Proposed offshore export cable are considered in this section of this chapter.
- The Proposed offshore export cable is considered unlikely to have the potential to result in a cumulative impact with the Array, other than in very close proximity to the Array. This is due to the nature and scale of the proposed offshore export cable works which are likely to be restricted in nature and associated only with cable laying. The total footprint is not available at present.
- At the time of writing this Array EIA Report, there was no EIA Report available for the Proposed offshore export cable corridor(s), but the activities and footprints of disturbance associated with its site preparation and construction phase are expected to be similar to those of other cable laying projects e.g. Easter Green Link 2.
- Activities associated with the site preparation and construction phase for the Proposed offshore export cable corridor(s) are expected to be of an equal or lesser extent than those represented by the MDS for the Array alone for all relevant topics. The impacts of cable installation and seabed preparation are likely to be reversible. The cumulative magnitude of impact of the Array with the proposed offshore export cable is therefore not expected to represent a material additional impact to that defined for the assessment of the Array alone for any assessment presented in the volume 2, chapters 7 to 20. Within this phase of development of the Array, site preparation and construction activities are anticipated to occur intermittently. They will be spread out across the full allotted timeframe with only a small proportion of the MDS footprint for this impact being affected at any one time. There may be some spatial overlap between the Array and the Proposed offshore export cable corridor(s), given their proximity, however this is considered to be of very short duration and limited in extent.
- The cumulative impact is therefore predicted to be of local spatial extent, short term duration (between 2030 and 2038), intermittent, and of high reversibility. It is predicted that the impact will affect the receptors directly. The magnitude is therefore considered to be low and the sensitivity of the receptors is as set out in volume 2, chapters 7 to 20. The overall significance of assessment is therefore considered to be minor adverse significance, which is not significant in EIA terms.
20.9. Part Two: Ecosystem Based Effects Assessment
20.9. Part Two: Ecosystem Based Effects Assessment
20.9.1. Overview
20.9.1. Overview
- An ecosystem is a community of living (biotic) organisms existing in conjunction with the non-living (abiotic) components of their environment. These biotic and abiotic components are linked together through nutrient cycles and energy flows (LibreTexts, 2022). In marine ecosystems biotic components include plankton, seaweed, benthic communities, fish, seabirds and marine mammals and abiotic components include air, salt water, seabed components and rock.
- Biodiversity is defined as the collection of genomes, species, and ecosystems occurring in a geographically defined region (National Research Council, Division on Earth, Life Studies, Commission on Geosciences, Resources and Committee on Biological Diversity in Marine Systems, 1995). The biodiversity within an ecosystem is the key indicator of the health of an ecosystem. A wider variety of species will cope better with external pressures than a limited number of species in large populations. Even if certain species are affected by climate change or human activities, the ecosystem as a whole may adapt and survive (European Commission, 2022).
- The purpose of this ecosystem-based assessment is to qualitatively assess the potential effects of the Array at the ecosystem level, to better understand how predator – prey relationships could be altered and how this could impact the functioning of the ecosystem. This is to address the advice raised by NatureScot during consultation that “increasingly there is a need to understand potential impacts holistically at a wider ecosystem scale in addition to the standard set of discrete individual receptor assessments. This assessment should focus on potential impacts across key trophic levels particularly in relation to the availability of prey species. This will enable a better understanding of the consequences (positive or negative) of any potential changes in prey distribution and abundance from the development of the wind farm on seabird and marine mammal (and other top predator) interests and what influence this may have on population level impacts”.
20.9.2. Ecosystem Baseline
20.9.2. Ecosystem Baseline
- This section provides a summary of the abiotic and biotic components of the marine ecosystem relevant to the Array, considering the topic specific study areas used for assessment.
- The Array will be located within the site boundary, located off the east coast of Scotland, approximately 80 km south-east of Aberdeen from the nearest point, and comprising an area of approximately 859 km2. Across the site boundary, the maximum water depth was recorded at 88.7 m Lowest Astronomical Tide (LAT), and the shallowest area was recorded at 63.8 m LAT. The seabed across the site boundary is relatively flat with a gentle slope downwards in an approximately north-west to south-east direction (Ocean Infinity, 2022). The average water depth across the site boundary is 74.47 m below LAT.
- The seabed within the site boundary consists primarily of sand, with some areas of gravel and occasional diamicton (poorly sorted mixed sediments). Gravel areas are more frequent in the north-west, with occasional diamicton also observed in this area.
- The geological morphology within the site boundary is varied and includes the following features:
- megaripples;
- sand waves;
- boulders (primarily in the north-west);
- recent marine soft sediment deposits; and
- deep channel structures (down to 60 m) with sedimentary infill (south-eastern corner).
- The benthic communities within the site boundary are characterised by polychaetes (particularly bristleworm Spiophanes bombyx), dead man’s fingers Alcyonium digitatum, and various echinoderms and bryozoans (such as hornwrack Flustra foliacea). Biomass between grab sampling sites was varied, with six major phyla identified: Echinodermata, Mollusca, Annelida, Arthropoda, Cnidaria and Bryozoa. Echinoderms comprised the majority of the biomass within the grab samples (65%), which is largely due to the purple heart urchin Spatangus purpureus and sea potato Echinocardium cordatum occurring at several grab sampling sites. The phyletic composition was dominated by annelids, mainly sand mason worm Lanice conchilega and S. bombyx. The phyletic composition of sessile colonial fauna was dominated by cnidarians and bryozoans, with cnidarians representing the highest number of taxa and bryozoans the highest number of colonies. The closest designated site (located approximately 25 km west of the Array) is the Firth of Forth Banks Complex Marine Protected Area (MPA) which is designated for ocean quahog Arctica islandica, offshore subtidal sand and gravels, shelf banks and mounds, moraines representative of the Week Bankie Key Geodiversity Area (volume 2, chapter 8).
- Table 20.15 Open ▸ provides a summary of the seven main broad subtidal habitats present with the site boundary. Details of the grab sample locations (e.g. S018) can be found in volume 2, chapter 8.
Table 20.15: Broad Habitat Types and Species
- The other species groups which are part of the biotic components of the ecosystem include fish, seabirds and marine mammals. These groups are considered further in sections 20.9.4 and 20.9.5.
20.9.3. The Marine Food Web
20.9.3. The Marine Food Web
- Trophic levels describe the hierarchical levels which organisms occupy in the food web. Primary producers, such as phytoplankton and seaweed, form the lowest trophic levels in marine food webs. They are consumed by primary consumers (herbivores) such as zooplankton, some crustaceans (e.g. copepods) and molluscs (e.g. clams, snails, mussels). Secondary consumers (carnivores or omnivores) such as fish larvae, Atlantic herring Clupea harengus (hereafter referred to as “herring”) and lesser sandeel Ammodytes marinus, and some crustaceans (e.g. crabs, shrimp) feed on primary consumers and primary producers. These species support tertiary consumers (carnivores), including some fish species, and cephalopods (e.g. octopus and squid species). Seabirds, along with marine mammals, large marine fish and elasmobranchs (sharks, skates and rays), are the top predators of the natural marine food web. An example of a marine food web which illustrates the interactions between the different trophic levels is presented in Figure 20.1.
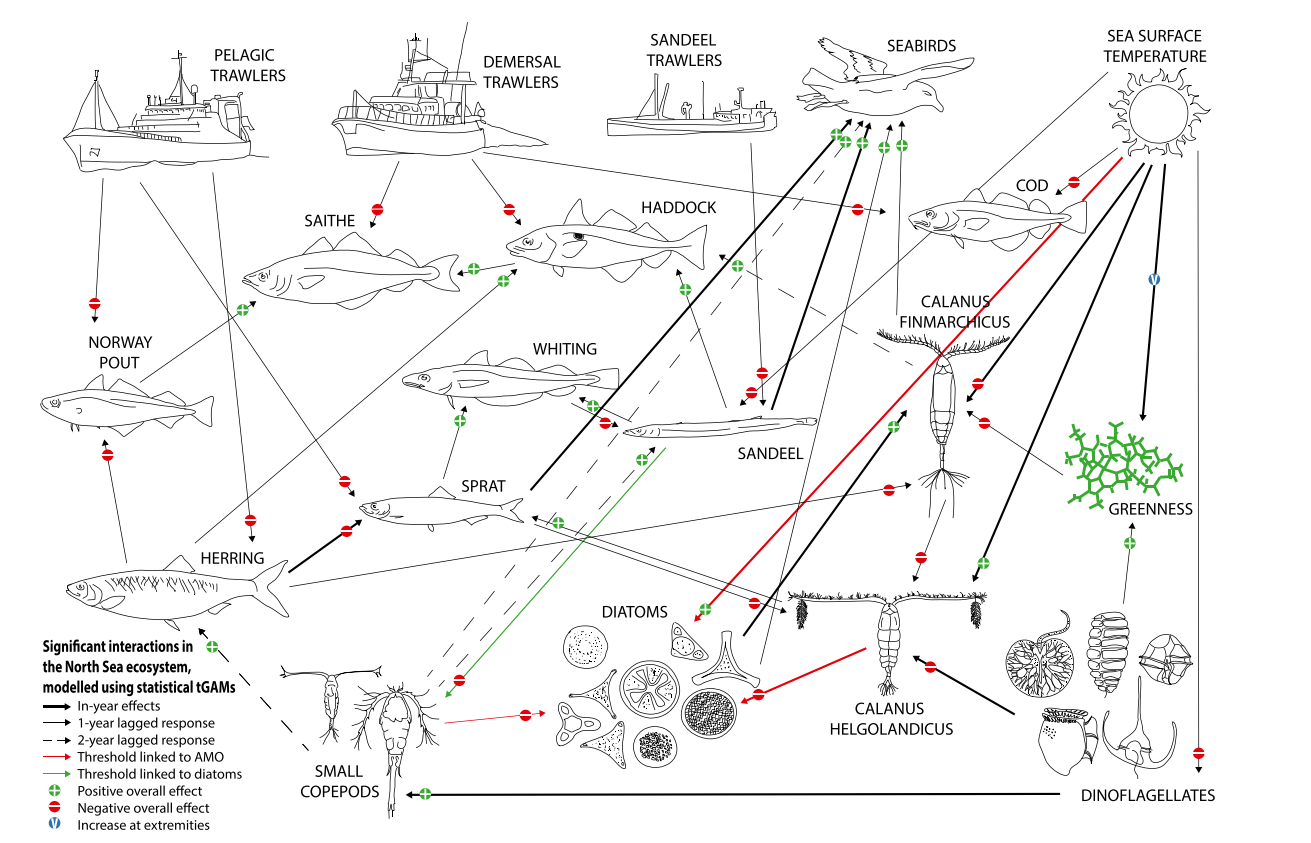
Figure 20.1: Significant Interactions Between Functional Groups and Drivers (from Lynam et al., 2017)
20.9.4. The Key Predator Species
20.9.4. The Key Predator Species
- Volume 2, chapters 9, 10 and 11, provide details on the fish, marine mammals and seabirds which are most abundant in the associated topic study areas and are the receptors most likely to be impacted by activities associated with all phases of the Array. From information on these receptor groups it is possible to ascertain which fish, seabird and marine mammal species are likely to be key predators in the marine ecosystem in this part of the central North Sea and within the study areas outlined in section 20.3.
Piscivorous fish
- The key marine predatory fish likely to utilise the marine environment within the site boundary are cod, haddock Melanogrammus aeglefinus, whiting Merlangius merlangus, plaice Pleuronectes platessa, saithe Pollachius virens and European hake Merluccius poutassou (hereafter referred to as “hake”). Piscivorous fish primarily feed on other fish species with these diet of these species including small forage such as sandeel, juvenile whiting and juvenile haddock. Several elasmobranch species are also likely to be present whose diet includes small forage food such as tope shark Galeorhinus galeus, spurdog Squalus acanthias, common skate Dipturus batis and rays.
- The migration route of diadromous fish species which also feed on small forage fish, and which are likely to pass through the site boundary during their migration (volume 3, appendix 9.1) are Atlantic salmon Salmo salar, sea trout Salmo trutta, sea lamprey Petromyzon marinus, European eel Anguilla anguilla, allis shad Alosa alosa and twaite shad Allosa fallax.
- Table 20.16 Open ▸ lists the key predator species and the prey they feed on. This shows that although sandeel, herring, mackerel Scomber scombrus and European sprat Sprattus sprattus (hereafter referred to as “sprat”) are components of most of the key predators’ diets, other fish and benthic fauna are also important in the diet of marine predatory fish.
Table 20.16: Key Predatory Fish Species and their Prey
Marine mammals
- The key marine mammal species which are most abundant within and therefore have the potential to be impacted by the Array are:
- harbour porpoise;
- bottlenose dolphin;
- white-beaked dolphin Lagenorhynchus albirostris;
- minke whale Balaenoptera acutorostrata ;
- humpback whale Megaptera novaeangliae; and
- grey seal.
- These species correspond to the marine mammal IEF identified in volume 2, chapter 10. The sensitivity of marine mammals to prey availability within the site boundary will be affected by how important this area is to each species and how sensitive they are to prey availability. This is discussed further in section 20.9.10.
- A summary of the dietary preferences of key marine mammal species within the marine mammal study area is presented in Table 20.17 Open ▸ . Further details of the most appropriate density values for marine mammals that have then been taken forward in the assessment are detailed in volume 3, appendix 10.2.
Table 20.17: Diet and Abundance of Key Marine Mammal Species
Seabirds
- The key seabird species which are most abundant (listed in abundance order) and most likely to be impacted by the Array (volume 2, chapter 11) are:
- black-legged kittiwake Rissa tridactyla (hereafter referred to as “kittiwake”);
- common guillemot Uria aalge (hereafter referred to as “guillemot”);
- razorbill Alca torda;
- Atlantic puffin Fratercula arctica (hereafter referred to as “puffin”);
- northern fulmar Fulmarus glacialis (hereafter referred to as “fulmar”); and
- northern gannet Morus bassanus (hereafter referred to as “gannet”).
- Seabird species diet and foraging behaviour determine the extent to which individual species are impacted and can respond to changes in prey availability. This is discussed further in section 20.9.10. A summary of their typical feeding strategies and prey species of key seabird species that have the potential to be impacted by the Array have been outlined in Table 20.18 Open ▸ .
Table 20.18: Diet and Feeding Strategies of Key Seabird Species
20.9.5. The Key Prey Species
20.9.5. The Key Prey Species
- The key fish and shellfish prey species likely to be present within the fish and shellfish study area, are the small shoaling forage fish sandeel, herring, mackerel and sprat. Volume 2, chapter 9 identified that these fish species are IEFs. The abundance of each of these species within the fish and shellfish study area and their relative importance to predators is discussed in the species summaries below.
Sandeel
- Sandeels, resembling small eels, predominantly feed on plankton of variable sizes, ranging from small plankton eggs up to larger, energy-rich copepods. In Scottish waters, there are five sandeel species, with the lesser sandeel and greater sandeel Hyperoplus lanceolatus being the most abundant in the vicinity of the site boundary. Lesser sandeel (hereafter referred to as “sandeel”) and Raitt’s sandeel Ammodytes marinus are listed as PMFs and listed as protected features within the Turbot Bank Nature Conservation MPA, which occurs within the fish and shellfish ecology study area.
- As well as being abundant in Scottish waters, sandeels are highly nutritious and therefore serve as the preferred prey item for several fish species, seabirds, seals, dolphins and whales. Sandeel represent an important link between the lower and upper levels of the marine food web, feeding on plankton and preyed upon by marine predators like cod, kittiwake and harbour porpoise (NatureScot, 2022).
- Sandeel have a close association with sandy structures into which they burrow. They are largely stationary after settlement and show a strong preference to specific substrate types. For the purposes of considering sandeel habitat suitability, gravelly sand (between 30% and 5% gravel), slightly gravelly sand (between 5% and 1% gravel) and sand (under 1% gravel) in the European Marine Observation and Data Network (EMODnet) substrate data were classified as preferred habitat and sandy gravel (between 30% and 80% gravel) as marginal habitat. The substrates classified as preferred and marginal habitats all have a sand to mud ratio of nine to one or higher. Where no shading from sunlight is present, the habitat in that area is considered unsuitable for sandeel (volume 3, appendix 9.1).
- As described in volume 2, chapter 9 and volume 3, appendix 9.1, through predicted distribution modelling by Langton et al. (2021) the whole site boundary has extremely low probability of sandeel presence. Abundance data from grab sampling and epibenthic trawls within the site boundary indicated that when sandeel were present, it was predominantly in the north-west section of the site boundary, which aligns with the marginal and preferred habitats for species within this section. Higher densities within the north-west section were found closer to the coasts or towards the Firth of Forth (volume 3, appendix 9.1).
Herring
- Herring is a small shoaling forage fish which is a commercially important pelagic fish. Commonly found across much of the North Sea, herring filter feeds on plankton and minute sea creatures, supplemented by small sprats and fry of other fish species (British Sea Fishing, 2022).
- Nursery grounds for herring are abundant across the east Scottish and Northumberland coastlines (Ellis et al., 2012). These areas serve as feeding grounds for post-larvae juveniles and sub-adults, before individuals reach sexual maturity and migrate further offshore (International Council for the Exploration of the Seas (ICES), 2006).
- Herrings are a key prey species for numerous fish, birds and marine mammals. Herring are listed on the Scottish Biodiversity List (SBL) and as a PMF and are therefore considered a high priority species for conservation actions in Scotland (Fauchald et al., 2011; Casini et al., 2004).
- Spawning for herring usually takes place in shallow areas between approximately 15 m and 40 m depth. Herring utilise specific benthic habitats during spawning (e.g. gravel, shells and small stones) with muddy sediments considered unsuitable due to increased egg mortality via asphyxiation as a result of coarse sediment of these environments blocking the pores of the eggs.
- Herring are considered hearing specialists with an increased sensitivity to underwater noise and are therefore vulnerable to injury or disturbance from activities which generate underwater noise, such as pile driving (volume 3, appendix 10.1).
- North Sea herring fall into a number of different ‘races’ or stocks, each with different spawning grounds, migration routes and nursery grounds (Coull et al., 1998). North Sea autumn-spawning herring have been divided into three, mainly self-contained stocks – the Buchan/Shetlands, Banks/Dogger and Bight/Downs herring groups, which show differences in spawning areas and spawning periods. The Buchan/Shetlands stock spawns off the Scottish and Shetlands coasts in August and September and is the most relevant to the Array as the closest to the site boundary and fish and shellfish ecology study area (volume 3, appendix 9.1).
- Herring spawning grounds are most accurately mapped using a combination of herring larval data and sediment particle size analysis (PSA), as recommended by Boyle and New (2018).
- Site-specific surveys illustrated that the overwhelming majority of (95%) of the site boundary has unsuitable sediment composition for herring spawning. The only four stations considered suitable for herring spawning were sparsely distributed in the north-west and centre of the site boundary (volume 3, appendix 8.1). Preferred habitats are located directly north of the site boundary, in line with spawning grounds detailed in Coull et al. (1998).
Mackerel
- Mackerel is a small, fast, predatory fish closely related to tuna Thunnini sp. which hunt in vast shoals for smaller fish and sandeel.
- As a vital prey species for larger fish, birds and marine mammals, mackerel are listed as PMFs in Scottish waters (NatureScot, 2020).
- Mackerel are migratory fish that are common throughout the UK, typically arriving in spring and early summer when they will feed actively before they migrate to warmer seas for the autumn and winter months for spawning. During this spawning period, their feeding activity diminishes significantly.
- Observations have been made that mackerel are arriving in UK waters earlier and leaving later every year, possibly as a result of rising sea temperatures. This has been linked to the complete absence of mackerel in areas around the south of the UK during the winter months.
- The absence of a swim bladder enables mackerel to swiftly adjust their depth, allowing them to maintain constant movement (British Sea Fishing, 2022).
- As described in volume 3, appendix 9.1, mackerel have low intensity nursery grounds which overlap with the site boundary, with no spawning grounds identified within or in proximity to the site boundary (Ellis et al., 2012). Mackerel spawning behaviour involves the release of eggs into the water column, where fertilisation also occurs (Walsh and Johnstone, 2006), indicating a low level of reliance on sedimentary habitats for spawning. Mackerel spawn over summer months from May to August. The presence of mackerel nursery grounds is not supported by outputs from Aires et al. (2014), with no modelled observations of “0 group fish” on the east coast of Scotland.
Sprat
- Sprat is a small foraging fish which is widespread across the UK, inhabiting water depths ranging from a few metres to approximately 100 m.
- Predominantly feeding on fish eggs, larvae, and plankton, sprat are a major part of the marine food chain in the North Sea. Sprat provide a vital food source for more or less all predatory fish species found in UK waters, as well as seabirds and marine mammals.
- As described in volume 3, appendix 9.1, sprat have an unidentified intensity spawning and nursery grounds over the majority of the site boundary.
20.9.6. How the Food System Works
20.9.6. How the Food System Works
- The transfer of energy moves up through the trophic levels of the food chain, starting at the bottom level where producers like phytoplankton and algae in the marine environment produce their own food by harnessing energy from the sun through the process of photosynthesis. Primary consumers, such as zooplankton, then feed on the phytoplankton to gain energy which is then transferred up each trophic level of the food chain.
- The marine environment typically follows a ‘wasp-waist’ trophic structure, where mid-trophic level species have lower diversity, compared to high diversity in both high and low trophic levels. These mid-trophic level species are crucial in the functioning of ecosystems (Rice, 1995). The main prey species found within the site boundary are sandeel, herring, mackerel and sprat. These fish link the lowest trophic level (e.g. phytoplankton) to the highest (e.g. marine mammals) (Mackinson and Daskalov, 2007; Feuchald et al., 2011; Lynam et al., 2017).
- Phenology plays an important role in how the food chain operates because many species have evolved elaborate behaviour and life history strategies that favour certain periods of the year for growth and reproduction and minimise the exposure of sensitive life stages in more stressful periods (Rubao et al., 2010). Any changes to phenology as a result of climate change can affect the lowest trophic levels and cause a cascade effect up through the food chain. For example, changes in sandeel populations will have a resulting effect on higher trophic species such as seabirds (Burthe et al., 2012; Lynam et al., 2017). This is further discussed in section 20.9.10.
- Section 20.9.4 describes the key fish, seabird and marine mammal predator species and their typical prey species. It can be noted that whilst the key prey species in section 20.9.5 are components of most predators’ diets, they vary in their importance. For example, kittiwake are more reliant on sandeel than the other key seabird species potentially present within the site boundary. Kittiwake would therefore be more sensitive to changes in sandeel distribution and availability. This is discussed further in section 20.9.10.
20.9.7. Future Ecosystem Baseline
20.9.7. Future Ecosystem Baseline
- The EIA Regulations require that a “a description of the relevant aspects of the current state of the environment (baseline scenario) and an outline of the likely evolution thereof without implementation of the Array as far as natural changes from the baseline scenario can be assessed with reasonable effort, on the basis of the availability of environmental information and scientific knowledge” is included within the Array EIA Report.
- If the Array does not come forward, an assessment of the ‘without development’ future baseline conditions has also been carried out and is described within this section.
Climate change effects
- The baseline environment for the physical and biological components of the ecosystem are subject to natural fluctuations over time. These changes will occur with or without the development of the Array due to natural variability. Therefore, it is important that when undertaking assessments of effects, any potential impacts must be considered within the context of the envelope of change that might occur over the timescale of the Array. Rising sea levels, increase sea temperatures and storminess are all likely to alter the future baseline conditions. In terms of physical processes, this is unlikely to have the effect of significantly altering tidal patterns and sediment transport regimes offshore at the site boundary. The return period of the wave climates would be altered (e.g. what is defined as a 1 in 50 year event may become a 1 in 20 year event) as deeper water would allow larger waves to develop. There is, however, uncertainty in the precise impacts climate change will have on prevailing wave climates within the North Sea and beyond.
- Sea surface temperatures (SSTs) around Scotland are strongly influenced by the atmosphere (heat flux) and ocean circulation (advection). Temperature variability in Scottish waters is provided regularly as part of the Scottish Ocean Climate Status Report (Hughes et al., 2018). Hughes et al. 2018 most recent report showed that Scottish waters (coastal and oceanic) have warmed by 0.05°C to 0.07°C per decade, calculate across the period of 1870 to 2016. Figure 20.2 shows an increasing trend in SST from 1893 to 2018 in all Scottish Marine Regions (SMRs) with trends for each region also ranging between 0.05°C and 0.07°C (Marine Scotland, 2024).
- Changes in temperature will affect the biological environment baseline (including benthic and intertidal ecology, fish and shellfish ecology, marine mammals and seabirds). Fish will be affected at all biological levels (cellular, individual, population, species, community and ecosystem) both directly and indirectly. For example, as sea temperatures rise, species adapted to cold water (e.g. herring and cod) will begin to disappear while warm water adapted species will become more established, creating a shift in the baseline. Changes in the stratification of water columns and plankton production may occur as a result of increased spring storms which may cause knock on effects through the food chain (Morison et al., 2019). The various changes that may occur as a result of climate change are uncertain and therefore it is difficult to predict the future baseline scenario with accuracy.
- Changes in ocean temperature, ocean acidification, water stratification and nutrient availability, as a result of climate change, are affecting the abundance and diversity of communities at all trophic levels (Walther, 2010). Effects have been identified over a variety of timescales. Short-term variability in environmental conditions impacts interactions between trophic levels and species (Howells et al., 2017). Limitations in prey availability can adversely affect top predators, with population level changes likely to occur over longer timescales, propagating up trophic levels with prolonged exposure (Frederiksen et al., 2006; Howells et al., 2017).
- The ability of fish species to move in response to temperature varies depending on a range of factors, including their physiological capacity to acclimatise and respond to the change as well as their degree of geographical attachment or how their prey respond. Where a species has a strong geographical attachment, the result can be a localised decline in species (Wright et al., 2020). Effects on prey species are further discussed in section 20.9.8.
- There is increased research into the effect of ocean acidification on fish physiology and early survival (Wright et al., 2020). As stated in paragraph 150, ocean acidification is a consequence of climate change due to chemical processes related to increased temperatures and increasing concentrations of carbon dioxide dissolving in seawater. The resulting decrease in pH is affecting phytoplankton which can inhibit shell generation of calcifying marine organisms and skeletal development of larval fish, with potential consequences for forage species (Riebesall et al., 2013). Impacts as a result of ocean acidification are however difficult to predict as species and population level due to the complexity of the different trophic levels within the food web (Heath et al., 2012).
- Understanding climate change impacts on top predators is fundamental to marine biodiversity conservation, due to their increasingly threatened populations and their importance in marine ecosystems (Olgeret et al., 2022). The long lifespans and large-scale mobility of top predators such as seabirds and marine mammals integrates information from the bottom to the top of the food chain and can serve as a sentinel of ecosystem change (Hazen et al., 2019). Burthe et al. (2014) conducted a long-term monitoring study (36 years) which found that the majority of a wide range of seabird species (including fulmar, kittiwake and puffin) demonstrated a negative response to increased SST in terms of their population size, breeding success and adult survival. Gannets were found to be less vulnerable which could be due to their higher flexibility in foraging behaviour (particularly in terms of exploiting fisheries discards as an alternative food source) although it is more likely due to a lack of data for this species (Burthe et al., 2014). Cliff nesting species, such as kittiwake and razorbill, may be more sensitive to nest failure as a result of high winds and storm surges caused by climate change (Newell et al., 2015).
- Overall, gannet are thought to be buffered from the impacts of climate change, mostly relating to their ability to access a wider variety of prey, but they may be sensitive to controls on fisheries discards (Johnston et al., 2021). Guillemot, kittiwake, puffin and razorbill abundances have been closely linked to the success of their prey, particularly sandeel (Burthe et al., 2014).
- Most marine mammal and climate change related studies to date have focused on effects of sea ice change however new studies are beginning to be published that consider the broader impacts of climate change on marine mammals. The main impacts are geographic range shifts, reduction in suitable habitats, food web alterations and increased prevalence of disease. Increased SSTs and resulting marine mammal range shifts are leading to novel interactions, increased predation risk and competition for species (Waggitt and Evans, 2020; Martin et al., 2023).
Figure 20.2: Sea Surface Temperature Trend from the ISST Data Product for the Observational Period (1893 to 2018) Averaged by SMRs and Offshore Marine Regions (OMR) (Marine Scotland, 2024)
Highly Pathogenic Avian Influenza (HPAI)
- Seabirds have been severely affected by avian influenza. The most recent HPAI outbreak began in 2021 and has now spread through more than 70 bird species. The virus has moved into species such as guillemot, razorbill, and kittiwake, with dead birds washing up on beaches in Wales, and along the eastern coasts of Scotland and England. At this stage, with little quantitative information, it is difficult to conclude to what extent population levels have been impacted by HPAI. Further information as to how HPAI has been considered in the site-specific surveys of the offshore ornithology study area can be found in volume 3, appendix 11.1.
Sandeel fishery closure
- As described in volume 3, appendix 9.1, the highest density of the sandeel population is focussed on the Wee Bankie (approximately 57 km west of the site boundary), however sandeel do range across much of the North Sea. In the early 1990s, there was a substantial industrial sandeel fishery on the Wee Bankie, Marr Bank and Berwick Bank sandbanks. By 1993, landings from this area had peaked at over 100,000 tonnes (Greenstreet et al., 2010b). In 2000, this industrial sandeel fishery was closed in response to concerns that the fishery was having a deleterious effect on sandeel stocks within the Forth and Tay SMR.
- In 2000, the first year of the closure of the Forth and Tay SMR sandeel fishery, high levels of recruitment, combined with a lack of any significant fishing activity resulted in an immediate and substantial increase in the biomass of sandeel on the Wee Bankie sandbank (Greenstreet et al., 2010b). However, between 2001 and 2010, sandeel biomass steadily declined to levels that were similar to those observed when the sandeel fishery was active (Greenstreet et al., 2010b). This was thought to be due to the absence of sustained recruitment, meaning that predation and other causes of natural mortality still exceeded population growth (Greenstreet et al., 2010b).
- As described in the Array Derogation Case due to the concerns about stock levels, a sandeel fishery ban was implemented in English waters from 2021 through 2023 for UK vessels (Horton, 2022). Following the initiation of a consultation by the Scottish Government in 2023, in January 2024, the Sandeel (Prohibition of Fishing) (Scotland) Order 2024 was established, closing sandeel fishing in Scottish waters from 2024 onwards. The decision considers the role of sandeel in the marine ecosystem(Scottish Government, 2024).
- Additionally, the UK government conducted a public consultation on spatial management measures for sandeel fishing in English waters of the North Sea. This consultation followed a 2021 call for evidence, revealing concerns about the impact of industrial fishing on the marine environment. Expert reports indicated that prohibiting sandeel fishing in the North Sea would benefit seabirds, other fish species, and marine mammals. Over 95% of respondents supported some form of prohibition, with a majority favouring the closure of all English waters. Consequently, the UK government decided to prohibit sandeel fishing within English waters of ICES Area 4 (North Sea) starting from 26 March 2024, before the next sandeel fishing season (Defra, 2024).
20.9.8. Existing Pressures on Prey Species
20.9.8. Existing Pressures on Prey Species
- Before assessing the potential effects of the Array on prey species at an ecosystem level, it is important to understand the existing pressures on each prey species.
- The North Sea is one of the most anthropogenically impacted marine ecosystems (Halpern et al., 2015; Emeis et al., 2015). Small shoal fish in mid-level trophic levels experience top-down pressure from commercial fisheries whilst bottom-up processes driven by temperature, have dominated changes to planktonic groups since the 1960s. These pressures propagate up and down the food chain, with mid-trophic fish linking the pressures between the upper and lower trophic levels (Lynam et al., 2017).
- Forage fish landings constitute approximately one-third of global landings of marine fish, not including losses from bycatch discards (Alder et al., 2008). Historically, sandeel have been targeted commercially for their oil and use as an animal feed and fertiliser. Despite being managed, the majority of sandeel stocks have experienced severe declines due to a combination of overfishing and the effects of climate change (NatureScot, 2022). In March 2024, the UK government introduced a prohibited sandeel fishing within English waters of ICES Area 4 (North Sea) (Defra, 2024). Further details of this sandeel fishery closure can be found in paragraph 157 to 160.
- As described in volume 3, appendix 9.1, herring are a commercially important pelagic fish in the North Sea which was targeted in the vicinity of the site boundary. The herring stock collapsed entirely in the 1970s as a consequence of overfishing (Scottish Herring, 2023). Since then, stocks have shown signs of recovery supported by a herring recovery plan implemented for the North Sea in 1996 and a ban on discards for pelagic fisheries, including for herring, from 2015. Active management is however still required to avoid a recurrence of the collapse (Dickey-Collas et al., 2010).
- The prey species present in the marine ecosystem within which the Array occurs, are also an important food source for larger fish. For example, plaice, cod, haddock, whiting, saithe, tope shark and spurdog all include prey forage species in their diet such as sandeel, herring, sprat and mackerel. Additionally, diadromous fish species are also likely to feed on these species. Volume 2, chapter 9, identified the following diadromous species are likely to migrate through the fish and shellfish ecology study area: Atlantic salmon, sea trout, sea lamprey, European eel, allis shad and twaite shad.
- As described in section 20.9.7, changes to the baseline environment as a result of climate change will have effects on marine fish species across all trophic levels. In terms of prey species, sandeel and herring are particularly vulnerable to the effects of climate change.
- Sandeel are one of the most important trophic links between plankton and top predators in North Sea ecosystems however climate driven changes to plankton and zooplankton have led to declines in the abundance and nutritional quality of sandeel since 2000 (Macdonald et al., 2015; Clausen et al., 2017; Wanless et al., 2018; MacDonald et al., 2019). This has caused knock-on effects up through the food chain.
- Sandeel are also impacted directly by climate change through their metabolic rate, which can in turn affect the success of their reproduction and increase their mortality rate (MCCIP, 2018; NatureScot, 2022). Increased temperatures have been observed to cause inhibited gonad development in sandeel, which means warmer seas can delay the spawning time and lead to reduced reproductive success (Wright et al., 2017). A key factor in sandeel larval success is synchrony between the larval hatching times and the spring zooplankton bloom. Adult sandeel feed on zooplankton in the spring and summer months; building up lipids to survive the winter period buried in sand when plankton production is lower. Increased temperatures lead to increased energy usage whilst overwintering, meaning less energy can be allocated to gonad development. (Boulcott and Wright, 2008; Wright et al., 2017). The delay to spawning time caused by inhibited gonad development can therefore lead to later larval hatch times, earlier zooplankton blooms and a resulting decrease in zooplankton available for sandeel to feed upon. This consequently can cause a reduction in sandeel growth and survivorship and ultimately low recruitment of the species (Réginer, Gibb and Wright, 2017).
- The life cycle of sandeel ties them to sandy sediments of a particular grain size which they burrow into at night and during the winter months. This means that their ability to move and redistribute to new suitable habitats in response to rising sea temperature relies on larval distribution (Macdonald et al., 2015).
- Herring are also constrained as demersal spawners, by their requirement to spawn at specific locations, depositing their sticky eggs on coarse sand, gravel, small stones and rocks (Wright et al., 2020).
20.9.9. Effects of the Array on Prey Species
20.9.9. Effects of the Array on Prey Species
- This section assesses the potential effects of the Array on prey species and any impacts on physical processes which may impact prey species indirectly by altering their availability to food sources such as plankton and zooplankton.
- Information to support this assessment has been extracted from the relevant receptor topic Array EIA Report chapters. Conclusions on LSE1 have also been extracted from these chapters. Each assessment of an effect focuses on the prey species most vulnerable to the impact and therefore represents the greatest potential impact.
Potential impacts on prey species
- Volume 2, chapter 9 identified that the following potential impacts as a result of the Array could result in the following potential impacts on fish and shellfish and benthic ecology:
- temporary habitat loss and disturbance;
- long-term habitat loss and disturbance;
- colonisation of hard structures;
- underwater noise impacting fish and shellfish receptors;
- underwater noise from the operation of floating wind turbines and anchor mooring lines impacting fish and shellfish receptors;
- increased SSCs and associated deposition; and
- effects to fish and shellfish receptors due to EMFs from subsea electrical cabling.
- Of the potential impacts, the first two were assessed as having minor adverse effects on marine fish (including prey species) which would not result in a significant change in prey species population. A summary of the assessment of these impacts is provided in the following sub-sections.
- The colonisation of hard structures has the potential to affect numbers of prey species and so is described in more detail, drawing on finds of volume 2, chapters 8, 9 and 10. While not likely to have a significant effect during the operation and maintenance phase, this impact could have an effect in the decommissioning phase if hard structures are left in-situ.
Temporary habitat loss and disturbance
- As discussed in volume 2, chapter 9, in general, mobile fish are able to avoid areas subject to temporal habitat disturbance. Of the key prey species, sandeel and herring are more sensitive to temporary habitat loss as they spawn on or near the seabed however recovery is expected to occur quickly as the sediment recovers post-construction and recolonisation occurs. Furthermore, the conditions in the fish and ecology study area are largely unsuitable for herring and sandeel habitats, and so there is limited disturbance to the spawning of these species. While there is a small overlap with herring spawning grounds, the impact is expected to be very limited due to the context of available favourable sediments habitat outside and across the fish and shellfish ecology study area.
Long term habitat loss and disturbance
- As discussed in volume 2, chapter 9, long term habitat loss and disturbance may arise due to the operation and maintenance phase of the Array. As with the potential impact of temporary habitat loss and disturbance, the most sensitive species are sandeel and herring. They are expected to recover quickly as the sediments recover following installation of Array infrastructure and adults and larvae recolonise the sandy sediments. Sandeel are particularly sensitive to long-term habitat loss and disturbance because of their specific habitat requirements (e.g. sandy sediments) for spawning and burrowing at night and through the winter. However, while sandeel are assessed to have medium sensitivity to this impact, the impact is expected to be limited in extent (particularly in the context of available habitats in the fish and shellfish ecology study area and the wider northern North Sea). Given the limited availability of favourable sediments within the site boundary, significant effects are not predicted.
- Herring habitat is largely unsuitable within the site boundary, and with spawning grounds existing outside the site boundary in the wider fish and shellfish ecology study area. There is, however, a small overlap with the herring spawning habitat and the fish and shellfish ecology study area. However, the area of herring spawning grounds affected by this impact is expected to be very limited (being limited to the site boundary only), in the context of available favourable sediments habitat outside and across the fish and shellfish ecology study area.
Colonisation of hard structures
- Volume 2, chapters 8, 9 and 10 discussed how the introduction of infrastructure within the Array may result in the colonisation of foundations, scour protection and cable protection. Since these hard structures are added to the areas typically characterised by soft, sedimentary environments, the resulting change of habitat type acts like an artificial reef. Anthropogenic structures on the seabed attract many marine organisms including benthic species normally associated with hard biological structures (e.g. blue mussel) (Karlsson et al., 2022). Additionally, man-made structures may also have direct effects on fish through their potential to act as fish aggregation devices (Peterson and Malm, 2006). While the reef effect can affect the existing biological soft sediment communities it can also have potentially beneficial effects on the marine ecosystem.
- The colonisation of new habitats may potentially lead to the introduction of INNS. This may have resulting impacts on benthic and fish and shellfish populations as a result of competition. A study into the spread of INNS by wind farm hard structure colonisation suggested the risk of this occurring was minor, and requires more research to fully understand, with implementation of precautionary built-in measures recommended to prevent spread where possible (Baulaz, et al., 2023). Potential adverse effects of the introduction of INNS are discussed further in volume 2, chapter 8.
- Artificial reefs can act as stepping-stones allowing organisms to colonise areas not typical of their species which can increase the connectivity between natural sub-populations (Coolen et al., 2017). The impacts of this can extend beyond the local scale of a single operation (e.g. the Array) with multiple adjacent offshore wind farms creating stepping stones across wider areas and creating a large-scale effect (Degraer et al., 2020). For example, the Array is close to three offshore wind farms: Seagreen 1 Offshore Wind Farm, Kincardine Offshore Wind Farm and Seagreen 1A Project (volume 2, chapter 15). This cumulative effect does not extend to benthic communities which are unlikely to be suited to the sedimentary habitats between projects and therefore will only colonise the hard structures of individual projects. Despite this, increased vessel presence could provide vectors and stepping stones for larval species and INNS. As species become established on and around the artificial hard structures, they can start producing larvae, with one study demonstrating that networks of oil and gas infrastructure in the North Sea could facilitate ecological connectivity by acting as stepping stones for larval connectivity (Henry et al., 2018) (volume 2, chapter 8). There is some evidence (although with uncertainties) that some fish and shellfish populations are likely to benefit from introduction of hard structures. See further details on the effects of the colonisation of hard structures on benthic and fish and shellfish ecology from paragraph 182 et seq.
Benthic ecology
- A review by Degraer et al. (2020) explained the process by which rapid colonisation can occur on all submerged parts of wind turbine components. Vertical zonation of species is usually observed with different species colonising the splash, inter-tidal, shallow and deeper subtidal zones (Degraer et al., 2020). Colonising communities on offshore installations are typically dominated by mussels, macroalgae and barnacles near the water surface, which essentially creates a new intertidal zone, while the community is dominated by filter feeding arthropods at intermediate depths, and by anemones at deeper locations (De Mesel et al., 2015; Karlsson et al., 2022). Colonisation of the hard substrates associated with the Array is therefore likely to result in an increase in biodiversity and a change compared to the baseline if no hard substrates were present (Lindeboom et al., 2011). In addition, the structural complexity of artificial substrates such as Offshore Substation Platform (OSP) foundations and floating wind turbine foundations may provide refuge as well as increasing feeding opportunities for larger and more mobile species. For example, Mavraki et al. (2020), demonstrated higher food web complexity associated with zones which had high accumulation of organic material (such as soft substrate or scour protection), suggesting potential reef effect benefits from the presence of artificial hard structures.
- Colonisation of hard structures may have indirect effects on the baseline communities and habitats identified within the Site boundary due to increased predation on and competition for the existing soft sediment species. These effects are difficult to predict, especially as monitoring to date has focused on the colonisation and aggregation of species close to the wind turbine foundations rather than broad scale studies.
- Some studies have also shown that the installation and operation of offshore wind farms has a negligible impact on the soft sediment environments. De Backer et al. (2020) found that the soft sediment benthic community underwent no drastic changes eight to nine years after the installation of C-power and Belwind Offshore Wind Farms in Belgium and that the species originally inhabiting the sandy substrate were still present and remained dominant in the offshore wind farms. Hutchinson et al. (2020) found that, during post-construction monitoring at the Block Island Wind Farm in the USA, no strong gradients of change in sediment grain size, enrichment or benthic macrofauna within 30 m to 90 m distance of the wind turbines was found. APEM (2022) found that at the Beatrice Offshore Wind Farm in the Moray Firth, colonisation of wind turbines resulted in zonation of the foundation itself and had little influence on the sedimentary habitat below. Across all wind turbines, plumose anemones Metridium senile and tube worms Spirobranchus sp. were the most abundant species, with the highest biomass at 40 m depth. Similarly, at the Hywind Scotland Pilot Park off the coast of Aberdeenshire, plumrose anemones and tube worms. dominated the bottom and mid-section of wind turbines, and a general increase of epifouling growth between 2018 and 2020 was recorded, indicating a source of food was present (Karlsson et al., 2022).
- The MDS assumes that up to 19.27 km2 of artificial hard substrate will be installed on the seabed within the Array benthic subtidal ecology study area (2.25% of the entire area). This comprises mooring lines and anchors on the seabed, OSP foundations, inter-array and interconnector cable protection and cable crossing protection, subsea junction boxes, and scour protection for mooring lines, anchors, OSP foundations, and subsea junction boxes. The floating wind turbine foundations represent up to 3.79 km2 of hard substrate which may be colonised within the water column. It is expected that these artificial hard structures will be colonised by epifaunal species local to the site boundary (volume 2, chapter 8).
- The ocean quahog IEF and phosphorescent sea pen IEF require a soft sedimentary habitat, and physical change to hard artificial or rock substratum would represent habitat loss for these species, which are highly vulnerable to this impact (Hill and Tyler-Walters, 2018).
- In contrast however, the dead man’s fingers IEF and the sea tamarisk IEF naturally live on hard substrates, including bedrock, rocks, boulders, shells, and man-made artificial hard structures (Budd, 2008, Wilson, 2002). Therefore, this impact does not represent a change from a preferred habitat to an unsuitable one for these IEFs, in comparison to the other IEFs. In addition, hydroids (such as sea tamarisk) are typically one of the first taxa to colonise new substrates (Boero, 1984). For example, a study on marine growth on the North Sea oil platform Montrose Alpha recorded eight species of hydroid (although none were sea tamarisk), present on the hard structures associated with the platform (Forteath et al., 1982).
Fish and shellfish ecology
- indirect effects on fish through the potential of the reef effect to bring about changes to food resources; and
- direct effect on fish through the potential to act as fish aggregation devices.
- The colonisation of epifaunal species on to the artificial hard structures of the Array may result in increased availability of prey species, which in turn may lead to increased numbers of fish and shellfish species utilising the hard substrate habitats.
- The introduction of hard structures such as foundations will likely lead to the colonisation of this substrate by fish and shellfish species. Primary colonisation may occur within hours or days by demersal and semi-pelagic species (Andersson, 2011). Colonisation has been seen to occur for a number of years following the initial construction, until a structured recolonised population is formed (Krone et al., 2013). The colonisation of these structures hence may attract fish from the surrounding areas to occupy the habitat with increased complexity, which will then increase the carrying capacity of the area (Andersson and Öhman, 2010; Bohnsack, 1989). The extent and nature of the colonisation of the new species will be determined by the dominant natural substrate character of the fish and shellfish ecology study area (largely muddy sand, sand, and slightly gravelly sand). For example::
- hard structures on an area of seabed are already characterised by rocky substrates, resulting in few new species but the ability to sustain a higher abundance (Andersson and Öhman, 2010); and
- hard structures on a soft seabed, may result in increased diversity of fish normally associated with rocky (or other hard bottom) habitats (Andersson et al., 2009). A new baseline species assemblage will be formed via recolonisation, and the original soft-bottom population will be displaced (Desprez, 2000).
- However, it was noted in volume 2, chapter 9, that the longest monitoring programme conducted to date at the Lillgrund Offshore Wind Farm in the Öresund Strait in southern Sweden, showed no overall decrease in fish numbers although redistribution towards the foundations within the offshore wind farm area was noticed for some species (i.e. cod, eel and eelpout) (Andersson, 2011). More species were recorded after construction than before, which is consistent with the hypothesis that localised increases in biodiversity may occur following the introduction of hard substrates in a soft sediment environment. However, there is uncertainty as to whether:
- artificial reefs facilitate recruitment in the local population; or
- the effects are simply a result of concentrating biomass from surrounding areas (Inger et al., 2009).
- Overall, results from earlier studies reported in the scientific literature did not provide robust data (e.g. some were visual observations with no quantitative data) that could be generalised to the effects of the addition of hard infrastructure on fish abundance in offshore wind farm areas (Wilhelmsson et al., 2010). More recent papers are, however, beginning to assess population changes and observations of recolonisation in a more quantitative manner (Bouma and Lengkeek, 2012; Krone et al., 2013), with hard structures consistently increasing species richness in the long term, but changing species composition towards a shellfish-dominated hard structures community, thus having an impact of local ecological function (Coolen, et al., 2020).
- Post construction fisheries surveys conducted in line with the Food and Environmental Protection Act licence requirements for the Barrow and North Hoyle offshore wind farms, found no evidence of fish abundance across these sites being affected, either beneficially or adversely, by the presence of the offshore wind farms (Centre for Environment, Fisheries and Aquaculture (Cefas), 2009; BOWind, 2008). These suggested that any effects, if seen, are likely to be highly localised, site dependent and while of uncertain duration, the evidence suggests effects are not necessarily adverse, although uncertainty does exist surrounding this issue (volume 2, chapter 9). Monitoring of fish populations in the vicinity of an offshore wind farm off the coast of the Netherlands indicated that the offshore wind farms acted as a refuge for at least part of the cod population (Lindeboom et al., 2011; Winter et al., 2010). Similarly, horse mackerel, mackerel, herring, and sprat have been found to utilise the new hard structures for spawning, or predation on the newly developed community (Glarou et al., 2020).
- The greatest potential benefit from the introduction of hard structures is likely to exist for crustacean species, such as crab and lobster. Evidence has been found that foundations can provide new potential sources of food, new potential habitat range and refuge areas and even successful hatchery and nursery grounds for several crab species (Linley et al., 2007; Hooper et al., 2014; BioConsult, 2006).
- Other shellfish species have the potential for great expansion of their normal habitats due to increased hard structures in areas of sandy habitat, as found in the fish and shellfish ecology study area. Krone et al. (2013) found that over a three-year period, almost the entire vertical surface of area of the platform piles had been colonised by three key species blue mussel, the amphipod Jassa spp. and anthozoans (mainly Metridium senile).
- In most cases, it is expected that diadromous fish are unlikely to utilise the increase in hard structures within the fish and shellfish ecology study area for feeding or shelter opportunities as they are only likely to be in the vicinity when passing through during migration to and from rivers located on the east coast of Scotland. Therefore, the reef effect is not anticipated to effect diadromous fish species numbers or behaviour. There is potential for impacts upon diadromous fish species resulting from increased predation by marine mammal species within offshore wind farms. Tagging of harbour seal and grey seal Dutch and UK wind farms provided significant evidence that the seal species were utilising wind farm sites as foraging habitats (Russel et al., 2014), specifically targeting introduced structures such as foundations. However, a further study using similar methods concluded that there was no change in seal behaviour within the offshore wind farm (McConnell et al., 2012), so it is not certain exactly to what extent seals utilise offshore wind developments and effects may be site-specific. Effects on marine mammals as a result of the colonisation of hard structures is discussed further in section 20.9.10.
- Research has shown that Atlantic salmon smolts spend little time in the coastal waters, and instead are very active swimmers in coastal waters, making their way to feeding grounds in the north soon after maturation (Gardiner et al., 2018; Newton et al., 2017; Newton et al., 2019; Newton et al., 2021) (see volume 3, appendix 9.1 for further detail on Atlantic salmon migration). Due to the evidence that Atlantic salmon tend not to forage in the coastal waters of Scotland, they are therefore at low risk of impact from increased predation from seals and other predators in the fish and shellfish ecology study area.
- Sea trout may be at higher risk of increased predation from seals than Atlantic salmon due to their higher usage of coastal environments. Given that sea trout are typically more coastal than Atlantic salmon, greater abundance would be expected further inshore than compared with the offshore waters of the site boundary (approximately 80 km offshore). Sea trout are generalist, opportunistic feeders with their diet comprising mainly of fish, crustaceans, polychaetes and surface insects with proportion of each of these prey categories varying dependent on season (Rikardsen et al., 2006; Knutsen et al., 2001). Due to the potential for increase in juvenile crustacean species and other shellfish species, which are possible prey items from sea trout, it is possible that foraging sea trout may be attracted to the hard structures introduced by installation of the Array. This attraction could in turn lead to increased predation of seal species upon sea trout species. However, there is little evidence at present documenting an increased abundance of sea trout around foundations (increases in fish abundance tend to be hard bottom dwelling fish species), therefore the above effect of increased prey items attracting sea trout is yet to be recorded. As sea trout abundance is typically greater inshore, it is unlikely that sea trout will spend time foraging around the foundations, and therefore there is a low risk of impact from increased predation from marine predators in the fish and shellfish ecology study area (volume 2, chapter 9).
Underwater noise impacting fish and shellfish receptors
- As discussed in volume 2, chapter 9, underwater noise may arise due to UXO clearance and piling for the installation of wind turbines and OSPs. This may cause direct and indirect impacts fish and shellfish receptors. However, this is unlikely to result in significant mortality due to the designed mitigation measures adopted as part of the Array (e.g. implementation of piling soft start and ramp up measures which will allow individuals to flee the area before noise levels reach a level at which injury may occur).
- Behavioural effects are expected over larger ranges. Some fish species (e.g. prat and herring) have special structures mechanically linking the swim bladder to the ear. Herring in particular are known to be particularly sensitive to underwater noise and have specific habitat requirements for spawning which makes them particularly vulnerable to impacts associated with construction related increases in underwater noise. However, due to the small proportion of undetermined intensity spawning grounds for herring within range of underwater sound levels, the effects are unlikely to result in a measurable impact on fish and shellfish receptors.
Underwater noise from the operation of floating wind turbines and anchor mooring lines impacting fish and shellfish receptors
- As discussed in volume 2, chapter 9 and volume 3, appendix 10.1, underwater noise has the potential to arise from wind turbine operation and movement of anchor mooring lines. This impact is relevant to the operation and maintenance phase and has the potential to cause direct and indirect impacts on fish and shellfish receptors.
- Studies have demonstrated that underwater noise from operational fixed wind turbines is only high enough to possibly cause a behavioural reaction in fish and shellfish species within metres from a wind turbine. In addition, noise generated by operational fixed wind turbines is of a low frequency and low sound pressure level (Andersson et al., 2011). Therefore, noise levels from operational wind turbines at a level where there is a potential effect on fish and shellfish receptors are considered highly unlikely to occur (Sigray and Andersson, 2011). These observations from earlier fixed offshore wind farms (with smaller wind turbines) are supported by modelling of the noise emissions from larger fixed offshore wind turbines, which demonstrate that the risk of injury or behavioural effects on fish and shellfish populations is negligible (SSER, 2022a). Putland (2022) and Risch et al. (2023) found that the operational noise of floating offshore wind is comparable to that of fixed bottom wind turbines, generating low level noise which is unlikely to cause significant disturbance effects to fish. Further details of these studies can be found in volume 2, chapter 9.
- It is acknowledged in volume 3 appendix 10.1 that underwater noise may occur due to mooring line slackening and tensioning which has the potential to produce transient ‘pinging’ or ‘snapping’ noises during the operation and maintenance phase of the Array (Liu, 1973). With specific reference to operational turbines, the distances and exposures of fish reported by various studies (as set out in volume 3, appendix 10.1) conclude that while sound levels would likely be audible, these would not be at a level sufficient to cause injury or behavioural changes to fish. This is due to the slight increase in SPL compared to the ambient noise measured before the construction of the wind farms and even when the highest increases in SPL was assumed (i.e. 20 to 25 dB re 1 μ Pa), these are unlikely to result in a measurable impact on fish and shellfish receptors.
Increased SSCs and associated deposition
- As stated in volume 2, chapter 9, the prey fish species most likely to be affected by sediment deposition are sandeel and herring because they spawn on the seabed. Sandeel have low intensity spawning and nursery grounds within the fish and shellfish ecology study area however sandeel eggs are likely to be tolerant to some level of sediment deposition due to the nature of the re-suspension and deposition within their natural high energy environment (Ellis et al., 2012). Therefore, effects on sandeel spawning populations are predicted to be limited. Sandeel populations are also sensitive to sediment type within their habitat, preferring coarse to medium sands and showing reduced selection or avoidance of gravel and fine sediments (Holland et al., 2005). This is as identified by the Feature Activity Sensitivity Tool (FeAST) tool as the pressure ‘siltation changes’ (low) which has identified that sandeel have medium sensitivity to this impact (Wright et al., 2000). Therefore, any increase in the fine sediment fraction of their habitat may cause avoidance behaviour until such time that currents remove fine sediments from the seabed, although modelled sediment deposition levels are expected to be highly localised and at very low levels.
- Herring occur mostly in pelagic habitats, but utilise benthic environments for spawning, and are known to prefer gravelly and coarse sand environments for this purpose, with low intensity nursery grounds present within the site boundary and low intensity spawning grounds nearby (Coull et al., 1998). With respect to the effects of sediment deposition on herring spawning activity, it has been shown that herring eggs may be tolerant of very high levels of SSC (Messieh et al., 1981; Kiorbe et al., 1981). However, detrimental effects may be seen if smothering occurs and the deposited sediment is not removed by the currents (Birklund and Wijsmam, 2005).
- The potential of an increase in SSCs may arise as a result of mooring lines or cables making contact with and moving on the seabed, disturbing seabed materials and causing scouring and increased SSCs within the water column. Any increase in SSCs and associated deposition will include native material only, and although comprises predominantly mobile sand material, the low rates of sediment transport, will ensure it is redeposited close by after a short period of suspension, thus not impacting significantly on seabed morphology. Any significant changes to the seabed morphology will not recover immediately, due to the low rates of sediment transport, however the evidence of mobile sediments implies any impacts will be fully recoverable after some time (volume 2, chapter 7).
Effects to fish and shellfish receptors due to EMFs from subsea electrical cabling
- As discussed in volume 2, chapter 9, the presence and operation of inter-array and interconnector cables within the fish and shellfish ecology study area may result in emission of localised EMFs which may affect some fish species. It is common practice to block the direct electrical field using conductive sheathing, meaning that the only EMFs that are emitted into the marine environment are the magnetic field and the resultant induced electrical field. Fish (particularly elasmobranchs) and shellfish species are able to detect applied or modified magnetic fields. However, the rapid decay of the EMF with horizontal and vertical distance (Bochert and Zettler, 2006) (i.e. within metres) minimises the extent of potential impacts. A study investigating the effect of EMFs on sandeel larvae spatial distribution found that there was no effect on the larvae (Cresci et al., 2022), and a prior study concluded the same for herring (Cresci et al., 2020).
Conclusions
- This section summaries the assessments from the topic specific chapters to inform the ecosystem effects assessment of the Array on prey species, to determine whether there will be any increases or decreases in predation and prey distribution and availability as a result of the Array.
- The impacts resulting from the lifetime of the Array (construction, operation and maintenance and decommissioning) which are relevant to prey species include temporary habitat loss and disturbance; long-term habitat loss and disturbance; colonisation of hard structures; underwater noise impacting fish and shellfish receptors; underwater noise from the operation of floating wind turbines and anchor mooring lines impacting fish and shellfish receptors; increased SSCs and associated deposition; and effects to fish and shellfish receptors due to EMFs from subsea electrical cables.
- The colonisation of hard structures has the potential to lead to increases in fish species through potential reef effect and fish aggregation. It is uncertain to what degree this may occur, however, any beneficial effects are predicted to be highly localised and not significant.
20.9.10. Effects of the Array on Predator Species
20.9.10. Effects of the Array on Predator Species
- Section 20.9.9 examined the impacts as a result of the Array which could have either positive or negative effects on the distribution of key prey species. This section assesses the sensitivity of fish, seabird and marine mammal predator species to prey availability and draws on the conclusions of section 20.9.9 to determine if there are any potentially significant effects on predators as a consequence of changes in prey availability. The likelihood of increased predation of key prey species as a result of the Array is considered highly unlikely due to the mobile nature of both prey and predator species and therefore has not been assessed further.
Piscivorous fish
- The typical prey species of the key predators (piscivorous fish) are listed in section 20.9.5 which shows these fish species have broad diets comprising not only of small fish but also benthic species including invertebrates, molluscs and crustaceans. This suggests, the fish predator species are likely to be less sensitive to the availability of the key prey species of sandeel, herring, mackerel and sprat.
- As discussed in section 20.9.9, adverse effects on prey species as a result of the Array were assessed to have adverse effects on marine fish (including prey species), which would not result in a significant change to prey species populations. The colonisation of hard structures has the potential to lead to localised increases in fish species through potential reef effect and fish aggregation. However, the assessments of effects concluded any increases would be localised and did not conclude that the Array would lead to a significant increase in prey species.
Marine mammals
- As discussed in volume 2, chapter 10, marine mammals are likely to profit from locally increased food availability and/or shelter and therefore have the potential to be attracted to forage within an offshore wind farm. While species such as harbour porpoise, minke whale, white-beaked dolphin, harbour porpoise and grey seal have been frequently recorded around offshore oil and gas structures, little is known about the how their distribution is linked to the reef effect or sheltering effect (Todd et al., 2016; Delefosse et al., 2018; Lindeboom et al., 2011). Acoustic results from a Towed Passive Acoustic Monitoring Device (T- POD) measurement within a Dutch wind farm found that relatively more harbour porpoises were found in the wind farm area compared to the two reference areas (Lindeboom et al., 2011, Scheidat et al., 2011). This study concluded that the presence within the wind farm area was due to increased food availability as well as the exclusion of fisheries and reduced vessel traffic in the wind farm (shelter effect). Further evidence suggesting that wind farms are used for foraging includes a study by Russell et al. (2014) where the movements of tagged harbour seals commonly exhibited grid-like movement patterns within two active wind farms in the North Sea. However, other studies have detected no statistical differences in the presence of harbour porpoises inside and outside a Danish wind farm (Brandt et al., 2009). Brandt et al. (2009) suggested, however, that a small increase in detections during the night at hydrophones deployed in close proximity to single wind turbines may indicate increased foraging behaviour near the monopiles. Whilst there is some mounting evidence of potential benefits of man-made structures in marine environment (Coolen et al., 2017), the statistical significance of such benefits and details about trophic interactions in the vicinity of artificial structures and their influence on ecological connectivity remain largely unknown (Elliott and Birchenough, 2022; Inger et al., 2009; McLean et al., 2022; Rouse et al., 2020).
- In terms of the reef effect, the assessment of effects concluded any increases would be localised and would not lead to a significant increase in prey species. For example, sandeel, a popular prey species for harbour porpoise, require specific sediment habitat conditions and are therefore unlikely to be attracted to the hard structures of offshore wind farm infrastructure.
- Marine mammals exploit a range of different prey items and can forage widely, sometimes covering extensive distances. As the potential impacts of construction on prey resources will be localised and largely restricted to the site boundary, only a small area will be affected when compared to the available foraging habitat in the North Sea. The fish and shellfish communities found within the fish and shellfish ecology study area (see volume 2, chapter 9) are characteristic of fish and shellfish assemblages in the northern North Sea. It is therefore reasonably to assume that, due to the highly mobile nature of marine mammals, there will be similar prey resources available in the wider area surrounding the site boundary.
- Despite this, foraging over greater distances could result in an energetic cost with the associated increased travel with this effect being particularly pertinent for harbour porpoise. Harbour porpoise has a high metabolic rate and only a limited energy storage capacity, which limits their ability to buffer against diminished food. Despite this, if animals do have to travel further to alternative foraging grounds, the impacts are expected to be largely short term in nature and reversible (i.e. elevated underwater noise would occur during site investigation surveys, geophysical surveys, vessel activity, UXO clearance, piling and other noise producing activities) and are likely to return to the area after the noise activity has ceased. Whilst the impact of elevated underwater noise from the operation of floating wind turbines and anchor mooring lines is long-term it is of highly local spatial extent and therefore of minor adverse significance. Injury or disturbance is discussed further in paragraphs 50 to 57.
- In volume 2, chapter 10 it was identified that minke whale have the potential to be particularly vulnerable to potential effects on sandeel, particularly if there is potential for reduced abundance. Studies analysing the stomach contents of minke whale found that in the North Sea this species is their key food resource, followed by clupeids Clupeidae and to a lesser extent mackerel (Robinson and Tetley, 2005; Tetley et al., 2008), see volume 3, appendix 10.2 for more details. However, as presented in volume 2, chapter 10, modelling by Langton et al. (2021) shows that the marine mammal study area has extremely low probability of sandeel presence, with areas where predicted density is high closer to the coasts or towards the Firth of Forth.
Seabirds
- Prey availability is one of the most important controls of species abundance and distribution in the higher trophic levels, including birds (Lynam et al., 2017; Mitchell et al., 2020). Reduced availability or shifts in the distribution of prey species means seabirds are having to travel further distances to forage for food. Fayet et al. (2021) conducted a study comparing the foraging behaviour of puffin populations across the north-east Atlantic and found that puffins from declining populations had to cover greater distances for foraging and had less energy-dense diets. Low prey availability close to the colonies, potentially resulting from climate or commercial fisheries effects, is also amplified by increased intra-specific and inter-specific competition which forces birds to forage further from their colonies.
- The extent to which seabirds respond to changes in prey availability is dependent on species. Generalist species, such as gulls, feed on a range of prey types and are therefore more resilient to these changes whereas specialist species, such as kittiwake, predominantly prey on small fish and struggle to adapt to changes in prey availability as easily (Furness and Tasker, 2000).
- Changes to prey distribution within the water column resulting from changes to stratification or temperature, will affect surface feeding species (e.g. kittiwake and terns) differently to water column feeding species (e.g. auks). Typically, water column feeding species can adapt better to changes in prey availability as they are not restricted to prey available in the upper 1 m to 2 m of the sea surface, as is the case for surface feeding species. The primary feeding strategies for key seabird species that have the potential to be impacted by the Array are detailed in Table 20.18 Open ▸ .
- The presence of sandeel has been linked to the reproductive success and survival of kittiwakes (Frederiksen et al., 2004, 2008; Carroll et al., 2017). During April and May, adult kittiwakes predominantly consume older sandeel (1+ year group), transitioning to juvenile (0 year group) sandeel in June and July while rearing chicks (Lewis et al., 2001). This dietary pattern aligns with the annual cycle of sandeel as 1+ sandeel group are active in the water column during spring and 0 year group, having newly metamorphosed from larvae to juveniles, are available from June. Both year groups then bury themselves over winter, surviving on the lipids they have accumulated during the spring months (Wright and Bailey, 1996). Sandeel stock levels have seen significant reductions as a result of climate change and commercial fisheries (as detailed in section 20.9.8) which may contribute to kittiwake declines (Caroll et al., 2017).
- In the Firth of Forth region, a decline in the average length-at-age of both the 0 year group and 1+ year group sandeel brought to puffin chicks on the Isle of May indicated a considerable decline in prey quality between 1973 and 2015. This trend is associated with reductions in kittiwake populations. It is estimated that the energy content of sandeel decreased by around 70% and 40% for 0 and 1+ sandeel groups, respectively, potentially leading to a significant change in the diet or behaviour of seabirds that rely on sandeel species (Wanless et al., 2018). The diet of chick-rearing kittiwakes, puffins, razorbills and shags was predominantly sandeel between 1973 and 2015 in the North Sea. More recently, a shift to sprat and herring has been observed in guillemots, razorbills and kittiwakes (Walness et al., 2018). Sprat feed and spawn repeatedly throughout spring and summer in coastal and offshore waters are therefore more readily available, which could account for this shift. As plunge divers, gannet predominantly feed on pelagic fish such as mackerel and sandeel or fisheries discards (Le Bot et al., 2019).
- Overall, the construction and operation of wind turbines may lead to changes in the behaviour, availability or distribution of prey species for seabirds. However, the majority of seabird species have large foraging ranges and a variety of target species (with the exception of little terns) ( Table 20.18 Open ▸ ) meaning they are able to adapt to short temporal changes in prey availability due to construction activities. This impact is further discussed in volume 2, chapter 11.
- The majority of marine fish species are expected to avoid habitat loss effects due to their greater mobility and recoverability post-construction. As discussed in section 20.9.9, sandeel are particularly vulnerable to long-term habitat and disturbance. However, the effects are unlikely to result in a measurable impact on fish and shellfish receptors.
- During the construction phase, as per volume 2, chapter 9, the impact to all fish and shellfish species is considered to be of negligible. Construction works will be spatially and temporally restricted, covering only a small portion of the site at any given time. Construction impacts are restricted to the duration of the construction phase, and once construction has finished, the adverse impacts will cease and any change on prey species will likely be reversed.
- During the operation and maintenance phase, as per volume 2, chapter 9, the impact to all fish and shellfish species is considered to be of negligible to minor adverse significance. Temporary habitat loss will occur as a result of the use of jack-up usage for operation and maintenance activities (10,500 m2 per year over the 35-year lifecycle), and also due to disturbance caused by reburial of inter-array and interconnector cables (1,222,400 m2 and 236,000 m2 per year, respectively). The maximum design scenario is for up to 51,411,500 m2 of temporary habitat loss/disturbance during the operation and maintenance phase. This equates to 5.99% of the total site boundary and therefore this represents a relatively small proportion of the fish and shellfish ecology study area. It should also be noted that only a small proportion of the total habitat loss/disturbance is likely to be occurring at any one time over the 35-year operation phase of the Array. During the operation and maintenance phase, changes to prey availability are expected to be minimal although as requested by NatureScot, this effect has been considered for this phase (volume 2, chapter 11). With the exception of little tern, the sensitivity of the VORs is considered to range between low to medium ( Table 20.19 Open ▸ ).
Table 20.19: Sensitivity of Receptors to Indirect Impacts from Construction/Decommissioning Noise
- It is challenging to separate the effects of different pressures, due to the complexity of how they interact and the combined impact they have on seabird populations, their environment and their prey at all scales. Although offshore wind farms can impact local seabird populations directly through displacement and collision, there may also be beneficial indirect impacts, such as the creation of artificial reefs and the resulting potential of an increase in prey availability (Coolen, 2017).
- Overall, gannet, herring gull and lesser black-backed gull are thought to be buffered from the impacts of climate change, mostly relating to their ability to access a wider variety of prey, but they may be sensitive to controls on fisheries discards (Johnston et al., 2021). Guillemot, kittiwake, puffin and razorbill abundances have been more closely linked to the success of their prey, which may make them more vulnerable to bottom-up climate change impacts (Burthe et al., 2014; Johnston et al., 2021). A reduction in prey quality and availability may also reduce the resilience of these species against storm events, which could lead to an increase in large-scale wrecks as climate change leads to an increase in extreme weather (Anker-Nilssen et al., 2017; Camphuysen et al., 1999; Heubeck et al., 2011; Morley et al., 2016). Cliff nesting species, such as kittiwake and razorbill, may also be sensitive to nest failure in high winds and storm surges (Newell et al., 2015).
- Climate change is considered to be the likely primary cause of decline in seabird populations in the future. It is believed that the absence of the Array would further delay the transition of the UK from reliance on fossil fuels and therefore further contribute towards climate change impacts and declining seabird populations.
Conclusions
- This section assessed whether there will be any changes to the key predator species as a result of the Array. This was achieved by assessing the sensitivity of the predator species to changes in prey availability and drawing on the conclusions of section 20.9.9 along with the findings of the relevant Array EIA Report chapters to determine if any changes to predator species are predicted. The following conclusions were made:
– broad range of prey species making them less sensitive to the availability of the key forage prey species (sandeel, herring, sprat and mackerel);
- marine mammals
– harbour porpoise
- may be more sensitive to disturbance due to the energetic cost associated with increased travelling, however, the impacts are expected to be short-term in nature and reversible;
– minke whale
- may be more sensitive to the any potential changes in the abundance or distribution of sandeel; and
- seabirds
– kittiwake are identified as being particularly sensitive to changes in prey availability of their favoured prey species, sandeel. Significant changes to prey species as a result of the Array are however not predicted due to the non-favourable habitats for sandeel within the fish and shellfish ecology study area.
20.10. Conclusion
20.10. Conclusion
- The inter-related effects for all topics have been assessed and are detailed above. It has been concluded that the inter-related effects across the lifetime of the Array will not result in combined effects of greater significance than the assessments presented for each of the individual phases and therefore the effect is not significant in EIA terms. It has also been concluded that multiple effects will not interact in a way that is likely to result in greater significance than those assessments presented for individual receptors. None of the potential impacts arising from the Array alone or in combination with other projects, will result in significant adverse effects on prey species and predator species.
- The ecosystem effects assessment concluded that whilst colonisation of hard structures, scour protection and cable protection has the potential to lead to localised increases in fish species through potential reef effects, any increases would be localised and are not expected to lead to a significant increase in prey species.
- Predator species most vulnerable to changes in prey availability arising from the Array impacts include harbour porpoise, minke whale and kittiwake. However, as significant changes to prey species as a result of the Array alone and in combination with other projects are not predicted, significant effects on the key predator species are also not predicted.
- It is concluded that there will be no adverse effects on seabirds arising from changes in the behaviour or availability of prey species for seabirds as a result of the Array. As outlined above, the majority of seabird species have a variety of target prey species and have large foraging ranges, meaning that they can forage for alternative prey species or move to other foraging areas if prey becomes temporarily unavailable due to construction activities.
20.11. References
20.11. References
Aires, C., González-Irusta, J.M. and Watret, R. (2014). Updating Fisheries Sensitivity Maps in British Waters. Scottish Marine and Freshwater Science Vol 5 No 10. Edinburgh: Scottish Government, 88pp. DOI: 10.7489/1555-1.
Andersson, M. H. (2011). Offshore Wind Farms – Ecological Effects of Noise and Habitat Alteration on Fish. PhD Thesis, Department of Zoology, Stockholm University.
Andersson, M. and Öhman, M. (2010). Fish and sessile assemblages associated with wind-turbine constructions in the Baltic Sea. Marine and Freshwater Research 61, 642-650.
Andersson, M.H., Berggren, M., Wilhelmsson, D. and Öhman, M.C. (2009). Epibenthic colonization of concrete and steel pilings in a cold-temperate embayment: a field experiment. Helgoland Marine Research, 63, 249-260.
Anker-Nilssen, T., Harris, M.P., Kleven, O. and Langset, M. (2017). Status, origin and population level impacts of Atlantic puffins killed in a mass mortality event in the southwest Norway early 2016. Seabird, 30, 1-14.
APEM. (2022). Beatrice offshore wind farm post-construction monitoring Year 2 (2021): Benthic grab survey report. Report on behalf of Beatrice Offshore Wind Farm Ltd. Available at: https://marine.gov.scot/sites/default/files/bowl_2021_post_construction_ornithology_monitoring_report_25_07_2023.pdf. Accessed on: 01 March 2024.
Arrigo, K. R., Van Dijken, G. L., Cameron, M. A., Van Der Grient, J., Wedding, L. M., Hazen, L., Leape, J., Leonard, G., Merkl, A., Micheli, F., Mills, M. M., Monismith, S., Ouellette, N. T., Zivian, A., Levi, M. and Bailey, R. M. (2020). Synergistic interactions among growing stressors increase risk to an Arctic ecosystem. Nature Communications, 11 (1). DOI:10.1038/s41467-020-19899-z.
Baulaz, Y., Mouchet, M., Niquil, N., & Lasram, F. B. R. (2023). An integrated conceptual model to characterize the effects of offshore wind farms on ecosystem services. Ecosystem Services, 60, 101513.
Benhemma-Le Gall, A., Graham, I. M., Merchant, N. D. and Thompson, P. M. (2021). Broad-Scale Responses of Harbor Porpoises to Pile-Driving and Vessel Activities During Offshore Windfarm Construction. Frontiers in Marine Science, 8. DOI:10.3389/fmars.2021.664724.
BioConsult (2006). Hydroacoustic Monitoring of Fish Communities at Offshore Wind Farms, Horns Rev Offshore Wind Farm, Annual Report 2005
Bochert, R., and Zettler, M.L. (2006). Effect of Electromagnetic Fields on Marine Organisms. In: Köller, J., Köppel, J., and Peters, W. (eds) Offshore Wind Energy. Springer, Berlin, Heidelberg, 223-34. Available at: https://doi.org/10.1007/978-3-540-34677-7_14. Accessed on: 28 March 2024.
Bohnsack, J. A. (1989). Are High Densities of Fishes at Artificial Reefs the Result of Habitat Limitation or Behavioural Preference? B. Mar. Sci., 44(2), pp. 631-645.
Boulcott, P. and Wright, P. (2008). Critical timing for reproductive allocation in a capital breeder: evidence from sandeels. Aquatic Biology, 3, 31-40.
Bouma, S. & W. Lengkeek. (2012). Benthic communities on hard substrates of the offshore wind farm Egmond aan Zee (owez). Including results of samples collected in scour holes. – Bureau Waardenburg, Culemborg. [report 11-205, NoordzeeWind report]
BOWind (2008). Barrow Offshore Wind Farm Post Construction Monitoring Report. First annual report. 15 January 2008, 60pp.
Boyle, G. and New, P. (2018). ORJIP Impacts from Piling on Fish at Offshore Wind Sites: Collating Population Information, Gap Analysis and Appraisal of Mitigation Options. Final report – June 2018. The Carbon Trust. United Kingdom. 247 pp.
Brandt, M.J., Diederichs, A. and Nehls, G. (2009). Harbour porpoise responses to pile driving at the Horns Rev II offshore wind farm in the Danish North Sea. Final report to DONG Energy. Husum, Germany, BioConsult SH.
British Sea Fishing (2022). Herring. Available at: https://britishseafishing.co.uk/herring/. Accessed on: 21 March 2024.
Buckstaff, K. C. (2004). Effects of watercraft noise on the acoustic behavior of bottlenose dolphins,Tursiops truncatus, in Sarasota Bay, Florida. Marine Mammal Science, 20 (4), pp.709-725. DOI:10.1111/j.1748-7692.2004.tb01189.x.
Burthe, S. J., Wanless, S., Newell, M. A., Butler, A., and Daunt, F. (2014). Assessing the vulnerability of the marine bird community in the western North Sea to climate change and other anthropogenic impacts. Marine Ecology Progress Series, 507, 277-295.
Camphuysen, C. J., Wright, P. J., Leopold, M., Huppop, O., and Reid, J. B. (1999). A review of the causes, and consequences at the population level, of mass mortalities of seabirds. ICES Cooperative Research Report No. 232.
Canning, S.J., Santos, M.B., Reid, R.J., Evans, P.G.H., Sabin, R.C., Bailey, N. and Pierce, G.J. (2008). Seasonal distribution of white beaked dolphins (Lagenorhynchus albirostris) in UK waters with new information on diet and habitat use. Journal of the Marine Biological Association of the UK, 88, pp 11591166.
Canning, S., Lye, G., Givens, L. and Pendlebury, C. (2013). Analysis of Marine Ecology Monitoring Plan Data from the Robin Rigg Offshore Wind Farm, Scotland (Operational Year 2). Natural Power Consultants, Dalry.
Carroll, M.J., Bolton, M., Owen, E., Anderson, G.Q.A., Mackley, E.K., Dunn, E.K. and Furness, R.W. (2017). Kittiwake breeding success in the southern North Sea correlates with prior sandeel fishing mortality. Aquatic Conservation: Marine and Freshwater Ecosystems, 27(6), pp.1164-1175.
Carter, M. I. D., Boehme, L., Cronin, M. A., Duck, C. D., Grecian, W. J., Hastie, G. D., Jessopp, M., Matthiopoulos, J., McConnell, B. J., Miller, D. L., Morris, C. D., Moss, S. E. W., Thompson, D., Thompson, P. M. and Russell, D. J. F. (2022). Sympatric Seals, Satellite Tracking and Protected Areas: Habitat-Based Distribution Estimates for Conservation and Management. Frontiers in Marine Science, 9. DOI:10.3389/fmars.2022.875869.
Casini. M, Cardinale. M, Arrhenius, F. (2004). Feeding preferences of herring (Clupea harengus) and sprat (Sprattus sprattus) in the southern Baltic Sea, ICES Journal of Marine Science, Volume 61, Issue 8, 2004, Pages 1267–1277, https://doi.org/10.1016/j.icesjms.2003.12.011.
Cefas (2009). Strategic Review of Offshore Wind Farm Monitoring Data Associated with FEPA Licence Conditions. Project ME1117. July 2009.
CES. (2006). Herring, Clupea harengus. Available at: https://www.ices.dk/about-ICES/projects/EURFP/EU%20Repository/ICES%20FIshMap/ICES%20FishMap%20species%20factsheet-herring.pdf. Accessed on: 21 March 2024.
Clausen, L. W., Rindorf, A., van Deurs, M., Dickey-Collas, M. and Hintzen N. T. (2017). Shifts in North Sea forage fish productivity and potential fisheries yield. Journal of Applied Ecology, 55(3), pp.1092-1101.
Convention on Biological Diversity (CBD) (2023). Ecosystem Approach. Available at: https://www.cbd.int/ecosystem/. Accessed on: 25 January 2024.
Coolen J.W.P. (2017). North Sea Reefs. Benthic biodiversity of artificial and rocky reefs in the southern North Sea. Unpublished PhD thesis, Wageningen University and Research.
Coolen, J. W., Van Der Weide, B., Cuperus, J., Blomberg, M., Van Moorsel, G. W., Faasse, M. A. and Lindeboom, H. J. (2020). Benthic biodiversity on old platforms, young wind farms, and rocky reefs. ICES Journal of Marine Science, 77(3), 1250-1265.
Coull, K.A., Johnstone, R., and S.I. Rogers (1998). Fisheries Sensitivity Maps in British Waters.
Cresci, A., Allan, B.J.M., Shema, S.D., Skiftesvik, A.B., and Browman, H.I. (2020). Orientation behaviour and swimming speed of Atlantic herring larvae (Clupea harengus) in situ and in laboratory exposures to rotated artificial magnetic fields. J. Exp. Mar. Biol. Ecol., 526, 151358. Available at: https://doi.org/10.1016/j.jembe.2020.151358. Accessed on: 28 March 2024.
Cresci, A., Perrichon, P., Durif, C.M., Sørhus, E., Johnsen, E., Bjelland, R., Larsen, T., Skiftesvik, A.B. and Browman, H.I., (2022). Magnetic fields generated by the DC cables of offshore wind farms have no effect on spatial distribution or swimming behaviour of lesser sandeel larvae (Ammodytes marinus). Marine Environmental Research, 176, 105609.
Damseaux, F., Siebert, U., Pomeroy, P., Lepoint, G., Das, K. (2021). Habitat and resource segregation of two sympatric seals in the North Sea. Science of The Total Environment, Volume 764, 142842, ISSN 0048-9697, https://doi.org/10.1016/j.scitotenv.2020.142842.
De Backer, A., Buyse, J. and Hostens, K. (2020). A decade of soft sediment epibenthos and fish monitoring at the Belgian offshore wind farm area. Environmental Impacts of Offshore Wind Farms in the Belgian Part of the North Sea: Empirical Evidence Inspiring Priority Monitoring, Research and Management. Series ‘Memoirs on the Marine Environment’. Royal Belgian Institute of Natural Sciences. Brussels, Belgium pp.79-113.
De Mesel, I., Kerckhof, F., Norro, A., Rumes, B. and Degraer, S. (2015). Succession and seasonal dynamics of the epifauna community on offshore wind farm foundations and their role as stepping stones for non-indigenous species. Hydrobiologia, 756 (1), pp.37-50. DOI:10.1007/s10750-014-2157-1.
DEFRA (2024). Consultation on spatial management measures for industrial sandeel fishing 1. Available at: https://www.gov.scot/publications/sandeel-prohibition-fishing-scotland-order-2024-final-business-regulatory-impact-assessment/. Accessed on: 27 March 2024.
Degraer, S., Carey, D.A., Coolen, J.W., Hutchison, Z.L., Kerckhof, F., Rumes, B. and Vanaverbeke, J., (2020). Offshore wind farm artificial reefs affect ecosystem structure and functioning. Oceanography, 33(4), pp.48-57.
Delefosse, M., Rahbek, M.L., Roesen, L. and Clausen, K.T. (2018). Marine mammal sightings around oil and gas installations in the central North Sea. Journal of the Marine Biological Association of the United Kingdom, 98(5), 993-1001.
DESNZ (2023a). Overarching National Policy Statement for Energy (NPS EN-1). Available at: https://assets.publishing.service.gov.uk/media/65bbfbdc709fe1000f637052/overarching-nps-for-energy-en1.pdf. Accessed on: 20 February 2024.
DESNZ (2023b). National Policy Statement for renewable energy infrastructure (NPS EN-3). Available at: https://www.gov.uk/government/publications/national-policy-statement-for-renewable-energy-infrastructure-en-3. Accessed on: 20 February 2024.
Desprez, M. (2000). Physical and biological impact of marine aggregate extraction along the French coast of the eastern English Channel: short and long-term post-dredging restoration. ICES Journal of Marine Science 57, 1428-1438.
ECOWind (2024). ECOWind Projects. Available at: https://ecowind.uk/projects/. Accessed on: 07 March 2024.
Edrén, S. M. C., Wisz, M. S., Teilmann, J., Dietz, R. and Söderkvist, J. (2010). Modelling spatial patterns in harbour porpoise satellite telemetry data using maximum entropy. Ecography, 33 (4), pp.698-708. DOI:10.1111/j.1600-0587.2009.05901.x.
Elliott, M. and Birchenough, S. N. R. (2022). Man-made marine structures – Agents of marine environmental change or just other bits of the hard stuff? Marine Pollution Bulletin, Volume 176. https://doi.org/10.1016/j.marpolbul.2022.113468.
Ellis, J.R., Milligan, S.P., Readdy, L., Taylor, N. and Brown, M.J. (2012) Spawning and nursery grounds of selected fish species in UK waters. Scientific Series Technical Report. Cefas Lowestoft, 147: 56 pp.
Emeis, K-C., van Beusekom, J., Callies, U., Ebinghaus, R., Kannen, A., Kraus, G., Kröncke, I., Lenhart, H., Lorkowski, I., Matthias, V. and Möllmann. (2015). The North Sea—a shelf sea in the Anthropocene. Journal of Marine Systems, 141, 18-33.
European Commission (1999). The Guidelines for the Assessment of Indirect and Cumulative Impacts as well as Impact Interactions. Available at: https://tethys.pnnl.gov/sites/default/files/publications/European-Commission-1999.pdf. Accessed on: 07 March 2024.
European Commission (2022). Why do we need to protect biodiversity? Available at: https://environment.ec.europa.eu/topics/nature-and-biodiversity_en. Accessed on: 07 March 2024.
Evans, P.G.H. (1990). European cetaceans and seabirds in an oceanographic context. Lutra, 33, 95–125.
Evans, P.G.H. and Waggitt, J.J. (2020) Impacts of climate change on marine mammals, relevant to the coastal and marine environment around the UK. MCCIP Science Review 2020, 421–455.
Faroese Safood, (2022). Saithe (Coley, Atlantic pollock). Available at: https://www.faroeseseafood.com/species/saithe-coley-atlantic-pollock/. Accessed on: 19 March 2024.
Fauchald, P., Skov, H., Skern-Mauritzen, M., Johns, D. and Tveraa, T. (2011). Wasp-waist interactions in the North Sea ecosystem. PLoS ONE, 6(7), e22729.
Fayet, A.L., Clucas, G.V., Anker‐Nilssen, T., Syposz, M. and Hansen, E.S. (2021). Local prey shortages drive foraging costs and breeding success in a declining seabird, the Atlantic puffin. Journal of Animal Ecology, 90(5), 1152-1164.
Fleming, A.H., Clark, C.T., Barlow, J. (2016). Humpback whale diets respond to variance in ocean climate and ecosystem conditions in the California Current. Global Change Biology. 22, 1214-1224. doi: 10.1111/gcb.13171.
Forney, K. A., Southall, B. L., Slooten, E., Dawson, S., Read, A. J., Baird, R. W. and Brownell Jr, R. L. (2017). Nowhere to go: noise impact assessments for marine mammal populations with high site fidelity. Endangered species research, 32, pp.391-413.
Frederiksen, M., Wanless, S., Harris, M.P., Rothery, P. and Wilson, L.J. (2004). The role of industrial fisheries and oceanographic change in the decline of North Sea black-legged kittiwakes. Journal of Applied Ecology, 41, pp.1129- 1139.
Frederiksen, M., Edwards, M., Richardson, A.J., Halliday, N.C. and Wanless, S. (2006). From plankton to top predators: bottom‐up control of a marine food web across four trophic levels. Journal of Animal Ecology, 75(6), 1259-1268.
Furness, R.W. and Tasker, M.L. (2000). Seabird-fishery interactions: quantifying the sensitivity of seabirds to reductions in sandeel abundance, and identification of key areas for sensitive seabirds in the North Sea. Marine Ecology Progress Series, 202, 253-264.
Gardiner, R., Main, R., Kynoch, R., Gilbey, J., and Davies, I., (2018). A needle in the haystack? Seeking salmon smolt migration routes off the Scottish east coast using surface trawling and genetic assignment. Poster presentation to the MASTS Annual Science Meeting 31 October – 2 November 2018.
Geelhoed, S. C. V., Authier, M., Pigeault, R. and Gilles, A. (2022). Abundance and distribution of cetaceans. In: OSPAR (2023): The 2023 Quality Status Report for the Northeast Atlantic [Online]. London: OSPAR Commission. Available at: https://oap.ospar.org/en/ospar-assessments/quality-status-reports/qsr-2023/indicator-assessments/abundance-distribution-cetaceans/. Accessed on: 20 March 2024.
Gerstein, E., Blue, J. and Forysthe, S. (2005). The acoustics of vessel collisions with marine mammals. Proceedings of OCEANS 2005 MTS/IEEE. IEEE.
Gilles, A., Authier, M., Ramirez-Martinez, N. C., Araújo, H., Blanchard, A., Carlström, J., Eira, C., Dorémus, G., Fernández-Maldonado, C., Geelhoed, S. C. V., Kyhn, L., Laran, S., Nachtsheim, D., Panigada, S., Pigeault, R., Sequeira, M., Sveegaard, S., Taylor, N. L., Owen, K., Saavedra, C., Vázquez-Bonales, J. A., Unger, B. and Hammond, P. S. (2023). Estimates of cetacean abundance in European Atlantic waters in summer 2022 from the SCANS-IV aerial and shipboard surveys. Final report published 29 September 2023 pp.64.
Glarou, M., Zrust, M., & Svendsen, J. C. (2020). Using artificial-reef knowledge to enhance the ecological function of offshore wind turbine foundations: Implications for fish abundance and diversity. Journal of Marine Science and Engineering, 8(5), 332.
Gordon, J., Gillespie, D., Potter, J., Frantzis, A., Simmonds, M. P., Swift, R. and Thompson, D. (2003). A Review of the Effects of Seismic Surveys on Marine Mammals. Marine Technology Society Journal, 37 (4), pp.16-34. DOI:10.4031/002533203787536998.
Gosch, M. (2017). The diet of the grey seal [Halichoerus grypus (Fabricius, 1791)] in Ireland and potential interactions with commercial fisheries. PhD Thesis, University College Cork.
Halpern, B.S., Frazier, M., Potapenko, J., Casey, K.S., Koenig, K., Longo, C., Lowndes, J.S., Rockwood, R.C., Selig, E.R., Selkoe, K.A. and Walbridge, S. (2015). Spatial and temporal changes in cumulative human impacts on the world’s ocean. Nature Communication, 6, 7615.
Hammond, P.S., Bearzi, G., Bjørge, A., Forney, K., Karczmarski, L., Kasuya, T., Perrin, W.F., Scott, M.D., Wang, J.Y., Wells, R.S. and Wilson, B. (2008). Phocoena. The IUCN Red List of Threatened Species 2008, e. T17027A6734992.
Hastie, G.D., Wilson, B., Wilson, L.J., Parsons, K.M., Thompson, P.M. (2004). Functional mechanisms underlying cetacean distribution patterns: hotspots for bottlenose dolphins are linked to foraging. Marine Biology (2004) 144: 397–403.
Hazen, E.L., Abrahms, B., Brodie, S., Carroll, G., Jacox, M.G., Savoca, M.S., Scales, K.L., Sydeman, W.J. and Bograd, S.J. (2019). Marine top predators as climate and ecosystem sentinels. Frontiers in Ecology and the Environment, 17(10), 565-574.
Heath, M.R., Neat, F.C., Pinnegar, J.K., Reid, D.G., Sims, D.W. and Wright, P. (2012). Review of climate change impacts on marine fish and shellfish around the UK and Ireland. Aquatic Conservation Marine and Freshwater Ecosystems, 22(3).
Heubeck, M., Aarvak, T., Isaksen, K, Johnsen, A, Petersen, I. K. and Anker-Nilssen, T. (2011). Mass mortality of adult Razorbills Alca torda in the Skagerrak and North Sea area, autumn 2007. Seabird, 24, pp.11-32.
Hill, J. M. and Tyler-Walters, H. (2018). Seapens and burrowing megafauna in circalittoral fine mud. In Tyler-Walters H. and Hiscock K. (eds) Marine Life Information Network: Biology and Sensitivity Key Information Reviews. Marine Biological Association of the United Kingdom. Plymouth, UK.
Hooper, T., Hattam, C., & Austen, M. (2017). Recreational use of offshore wind farms: Experiences and opinions of sea anglers in the UK. Marine Policy, 78, 55-60.
Horton, H. (2022). UK may ban sandeel fishing in move to save threatened seabirds. The Guardian.. Available at: https://www.theguardian.com/environment/2022/dec/16/uk-may-ban-sandeel-fishing-in-move-to-save-threatened-seabirds. Accessed on 27 March 2024.
Howells, R.J., Burthe, S.J., Green, J.A., Harris, M.P., Newell, M.A., Butler, A., Wanless, S. and Daunt, F. (2017). From days to decades: short-and long-term variation in environmental conditions affect offspring diet composition of a marine top predator. Marine Ecology Progress Series, 583, pp.227-242.
Hughes, S.L., Hindson, J., Berx, B., Gallego, A. and Turrell, W.R. (2018) Scottish Ocean Climate Status Report 2016. Scottish Marine and Freshwater Science Vol 9 No 4, 167pp. DOI: 10.7489/12086-1.
Hutchinson, Z.L., Bartley, M., Degraer, S. and English, P. (2020). Offshore wind energy and benthic habitat changes: lessons from Block Island Wind Farm. Oceanography, 33(4), 58-69.
IEMA (2016). IEMA Environmental Impact Assessment Guide to Delivering Quality Development. Available at: https://www.iema.net/download-document/7014. Accessed on: 07 March 2024.
IJsseldijk, L., Brownlow, A., Davison, N., Deaville, R., Haelters, J., Keijl, G., Siebert, U. and Ten Doeschate, M. (2018). Spatiotemporal trends in white-beaked dolphin strandings along the North Sea coast from 1991–2017. 61: 153-163.
Inger, R., Attril, M.J., Bearhop, S., Broderick, A.C., Grecian, W.J., Hodgson, D.J., Mills, C., Sheehan, E., Votier, S.C., Witt, M.J., and Godley, B.J. (2009). Marine Renewable Energy: Potential Benefits to Biodiversity? An Urgent Call for Research. Journal of Applied Ecology, 46, 1145-1153.
Johnston, D.T., Humphreys, E.M., Davies, J.G. and Pearce-Higgins, J.W. (2021). Review of climate change mechanisms affecting seabirds within the INTERREG VA area. Report to Agri-Food and Biosciences Institute and Marine Scotland Science as part of the Marine Protected Area Management and Monitoring (MarPAMM) project.
Karlsson, R., Tivefälth, M., Duranović, I., Martinsson, S., Kjølhamar, A. and Murvoll, K. M. (2022). Artificial hard-substrate colonisation in the offshore Hywind Scotland Pilot Park. Wind Energy Science, 7 (2), pp.801-814. DOI:10.5194/wes-7-801-2022.
Kastelein, R. A., Helder-Hoek, L., Booth, C., Jennings, N. and Leopold, M. (2019). High Levels of Food Intake in Harbor Porpoises (Phocoena phocoena): Insight into Recovery from Disturbance. Aquatic Mammals, 45 (4), pp.380-388. DOI:10.1578/am.45.4.2019.380.
Knutsen, J., Knutsen, H., Gjøsæter, J. & Jonsson, B. (2001). Food of anadromous brown trout at sea. Journal of Fish Biology. 59. 533 – 543. 10.1111/j.1095-8649.2001.tb02359.x.
Krone, R. Gutowa, L. Joschko, TJ. Schröder, A. (2013). Epifauna dynamics at an offshore foundation Implications of future wind power farming in the North Sea. Marine Environmental Research, 85, 1-12
Lacey, C., Gilles, A., Börjesson, P., Herr, H., Macleod, K., Ridoux, V., Santos, M. B., Scheidat, M., Teilmann, J., Sveegaard, S., Vingada, J., Viquerat, S., Øien, N. and Hammond, P. S. (2022). Modelled density surfaces of cetaceans in European Atlantic waters in summer 2016 from the SCANS-III aerial and shipboard surveys. SCANS-III project report 2. University of St Andrews. UK pp.31.
Langton, R., Boulcott, P. and Wright, P.J. (2021). A verified distribution model for the lesser sandeel Ammodytes marinus. Mar Ecol Prog Ser 667:145-159. doi: https://doi.org/10.3354/meps13693.
Le Bot, T., Lescroel, A., Fort, J., Péron, C., Gimenez, O., Provost, P., and Gremillet, D. (2019). Fishery discards do not compensate natural prey shortage in Northern gannets from the English Channel. Biological conservation, 236, pp.375- 384.
Lewis, S., Wanless, S., Wright, P.J., Harris, M.P., Bull, J. and Elston, D.A. (2001). Diet and breeding performance of black-legged kittiwakes Rissa tridactyla at a North Sea colony. Marine Ecology Progress Series, 221, 277-284.
LibreTexts (2022). Ecosystem Dynamics. Available at: https://bio.libretexts.org/Bookshelves/Introductory_and_General_Biology/Book%3A_General_Biology_(Boundless)/46%3A_Ecosystems/46.01%3A__Ecology_of_Ecosystems/46.1A%3A_Ecosystem_Dynamics. Accessed on: 07 March 2024.
Lindeboom, H. J., Kouwenhoven, H. J., Bergman, M. J. N., Bouma, S., Brasseur, S., Daan, R., Fijn, R. C., de Haan, D., Dirksen, S., van Hal, R., Hille Ris Lambers, R., ter Hofstede, R., Krijgsveld, K. L., Leopold, M. and Scheidat, M. (2011). Short-term ecological effects of an offshore wind farm in the Dutch coastal zone; a compilation. Environmental Research Letters, 6 (3). DOI:10.1088/1748-9326/6/3/035101.
Linley, E.A.S., Wilding, T.A., Black, K., Hawkins, A.J.S. and Mangi S. (2007). Review of the Reef Effects of Offshore Wind Farm Structures and their Potential for Enhancement and Mitigation. Report from PML Applications Ltd and the Scottish Association for Marine Science to the Department for Business, Enterprise and Regulatory Reform (BERR), Contract No: RFCA/005/0029P
Liu, F. (1973). Snap loads in lifting and mooring cable systems induced by surface wave conditions. Naval Civil Engineering Lab Port Hueneme Ca.
Lynam, C.P., Llope, M., Möllman, C., Helaouët, P., Bayliss-Brown, G.A. and Stenseth, N.C. (2017). Interaction between top-down and bottom-up control in marine food webs. Proceedings of the National Academy of Sciences of the United States of America. 114 (8), 1952-1957. DOI 10.1073/pnas.1621037114.
MacDonald, A., Heath, M., Edwards, M., Furness, R., Pinnegar, J.K., Wanless, S., Speirs, D. and Greenstreet, S. (2015). Climate driven trophic cascades affecting seabirds around the British Isles. Oceanography and Marine Biology - An Annual Review, 53, pp.55-80.
MacDonald, A., Spiers, D.C., Greenstreet, S.P.R., Boulcott, P. and Heath, M.R. (2019). Trends in Sandeel Growth and Abundance off the East Coast of Scotland. Frontiers in Marine Science, 6, pp.201.
MacLeod, C.D., S.M. Bannon, G.J. Pierce, C. Schweder, J.A. Learmonth, J.S. Herman and R.J. Reid (2005). Climate change and the cetacean community of north-west Scotland. Biological Conservation 124 (4): 477-483.
Marine Directorate Licensing Operations Team (2023). Scoping Opinion for the Ossian Array. Available at: https://marine.gov.scot/sites/default/files/ossian_array_-_scoping_opinion.pdf. Accessed on: 16 February 2024.
Marine Scotland (2024). Temperature. Available at: https://marine.gov.scot/sma/assessment/temperature#results. Accessed on: 27 March 2024.
Martin, E., Banga, R. and Taylor, N.L. (2023). Climate change impacts on marine mammals around the UK and Ireland. MCCIP Science Review. 22.
Mavraki, N., Degraer, S., Moens, T. and Vanaverbeke, J. (2020). Functional differences in trophic structure of offshore wind farm communities: a stable isotope study. Marine Environmental Research, 157, 104868.
MCCIP (2018). Climate change and marine conservation: Sandeel and their availability as seabird prey. (Eds. Wright P, Regnier T, EerkesMedrano D and Gibb F) MCCIP, Lowestoft, 8pp. doi: 10.14465.2018.ccmco.006-sel.
McConnell, B., Lonergan, M. and Dietz, R. (2012). Marine Estate Research Report Interactions between seals and offshore wind farms. The Crown Estate
Mclean, D., Cerqueira, F., Luciana, B., Jessica, M., Karen, S., Marie-Lise, S., Matthew, B., Oliver, B., Silvana, B., Todd, B., Fabio, B., Ann, C., Jeremy, C., Scott, C., Pierpaolo, C., Joop, J., Michael, F., Irene, F., Ashley, G., Bronwyn, T. M. (2022). Influence of offshore oil and gas structures on seascape ecological connectivity. Global Change Biology. 32. 10.1111/gcb.16134.
McWhinnie, L. H., Halliday, W. D., Insley, S. J., Hilliard, C. and Canessa, R. R. (2018). Vessel traffic in the Canadian Arctic: Management solutions for minimizing impacts on whales in a changing northern region. Ocean & Coastal Management, 160, pp.1-17. DOI:10.1016/j.ocecoaman.2018.03.042.
Mitchell, M.D. and Harborne, A.R. (2020). Non-consumptive effects in fish predator–prey interactions on coral reefs. Coral Reefs, 39(4), 867-884.
Morison F, Harvey E, Franzè G and Menden-Deuer S (2019) Storm-Induced Predator-Prey Decoupling Promotes Springtime Accumulation of North Atlantic Phytoplankton. Front. Mar. Sci. 6:608. doi: 10.3389/fmars.2019.00608.
Morley, T.I., Fayet, A.L., Jessop, H., Veron, P., Veron, M., Clark, J. and Wood, M.J. (2016). The seabird wreck in the Bay of Biscay and the Southwest Approaches in 2014: a review of reported mortality. Seabird, 29, 22-38
Muto, M., Helker, V., Angliss, R., Allen, B., Boveng, P., Breiwick, J., Cameron, M., Clapham, P., Dahle, S. and Dahlheim, M. (2018). Draft 2018 Alaska marine mammal stock assessments. US Dep. Commer. NOAA Tech. Memo. NMFS-AFSC-XXX. Published for public review and comment on September, 18, pp.2018.
Nabe-Nielsen, J., Tougaard, J., Teilmann, J. and Sveegaard, S. (2011). Effects of wind farms on harbour porpoise behaviour and population dynamics. Danish Centre for Environment and Energy, Aarhus University. Aarhus, Denmark pp.48 pp.
National Research Council, Division on Earth, Life Studies, Commission on Geosciences, Resources and Committee on Biological Diversity in Marine Systems (1995). Understanding marine biodiversity. National Academies Press.
NatureScot, (2020). Priority Marine Features in Scotland’s seas – Habitats. NatureScot, Edinburgh.
NatureScot, (2022). Sandeel. Available at: https://www.nature.scot/plants-animals-and-fungi/fish/sea-fish/sandeel. Accessed on: 21 March 2024
NatureScot (2024). Minke whale. Available at: https://www.nature.scot/plants-animals-and-fungi/mammals/marine-mammals/minke-whale. Accessed on: 19 March 2024.
Newton, M. Honkanen, H. Lothian, A. and Adams, C (2019). The Moray Firth Tracking Project – Marine Migrations of Atlantic Salmon (Salmo salar) Smolts. Proceedings of the 2019 SAMARCH Project: International Salmonid Coastal and Marine Telemetry Workshop.
Newton, M., Main, R. and Adams, C. (2017). Atlantic Salmon Salmo salar smolt movements in the Cromarty and Moray Firths, Scotland. LF000005-REP-1854, March 2017.
Newton, M., Barry, J., Lothian, A., Main, R. A., Honkanen, H., McKelvey, S. A., Thompson, P., Davies, I., Brockie, N., Stephen, A., O’Hara Murray, R., Gardiner, R., Campbell, L., Stainer, P., & Adams, C. (2021). Counterintuitive active directional swimming behaviour by Atlantic salmon during seaward migration in the coastal zone. ICES Journal of Marine Science, 78(5), 1730–1743. https://doi.org/10.1093/icesjms/fsab024. Available on: 19 March 2024.
NOAA Fisheries (2024) Humpback whale. Available at: https://www.fisheries.noaa.gov/species/humpback-whale/overview. Accessed on: 20 March 2024.
Nowacek, S. M., Wells, R. S. and Solow, A. R. (2001). Short‐term effects of boat traffic on bottlenose dolphins tursiops truncatus, in Sarasota Bay, Florida. Marine Mammal Science, 17 (4), pp.673-688. DOI:10.1111/j.1748-7692.2001.tb01292.x.
Ocean Infinity (2022). Geophysical and Environmental Survey: E1E. Document number: 103995-SSE-MMT-SUR-REP-INTRE (Rev03).
Orgeret F, Thiebault A, Kovacs KM, Lydersen C, Hindell MA, Thompson SA, Sydeman WJ, Pistorius PA. (2022). Climate change impacts on seabirds and marine mammals: The importance of study duration, thermal tolerance and generation time. Ecology Letters. 25(1), 218-239. doi: 10.1111/ele.13920.
Paxton, C.G.M., Scott-Hayward, L., Mackenzie, M., Rexstad, E. and Thomas, L. (2016). Revised Phase III Data Analysis of Joint Cetacean Protocol Data Resources. JNCC Report No. 517, JNCC, Peterborough.
Petersen, J.K. and Malm, T. (2006). Offshore windmill farms: threats to or possibilities for the marine environment. AMBIO: A Journal of the Human Environment, 35(2), 75-80.
PINS (2018). Advice Note Nine: Rochdale Envelope. Available at: https://infrastructure.planninginspectorate.gov.uk/legislation-and-advice/advice-notes/advice-note-nine-rochdale-envelope/. Accessed on 20 February 2024.
PrePARED (2022). Annual Report 2022. Available at: https://owecprepared.org/wp-content/uploads/2023/05/PrePARED-Annual-Report-2022-FINAL.pdf. Accessed on: 28 February 2024.
Raoux, A., Tecchio, S., Pezy, J.-P., Lassalle, G., Degraer, S., Wilhelmsson, D., Cachera, M., Ernande, B., Le Guen, C., Haraldsson, M., Grangeré, K., Le Loc’H, F., Dauvin, J.-C. and Niquil, N. (2017). Benthic and fish aggregation inside an offshore wind farm: Which effects on the trophic web functioning? Ecological Indicators, 72, pp.33-46. DOI:10.1016/j.ecolind.2016.07.037.
Régnier, T., Gibb, F.M. and Wright, P.J. (2017). Importance of trophic mismatch in a winter-hatching species: evidence from lesser sandeel. Marine Ecology Progress Series, 567, 185-197.
Reidy, R.D., Lemay, M.A., Innes, K.G., Clemente-Carvalho, R.B.G., Janusson, C., Dower, J.F., Cowen, L.L.E., Juanes, F. (2022). Fine-scale diversity of prey detected in humpback whale feces. Ecology and Evolution. 12(12): e9680. doi: 10.1002/ece3.9680.
Riebesall, U., Gattuso, J., Thinsgstad, T.F. and Middelburg, J.J. (2013). Preface arctic ocean acidification: Pelagic ecosystem and biogeochemical responses during a mesocosm study. Biogeosciences, 10(8), pp.5619-5626.
Rikardsen, A.H., Amundsen, P-A., Knudsen, R. Sandring, S. (2006). Seasonal marine feeding and body condition of sea trout (Salmo trutta) at its northern distribution. ICES Journal of Marine Science, Volume 63, Issue 3, 2006, Pages 466–475.
Robinson, K.P. and Tetley, M.J., (2005). Environmental factors affecting the fine-scale distribution of minke whales (Balaenoptera acutorostrata) in a dynamic coastal ecosystem. ICES Annual Science Conference, Aberdeen, Scotland, 20–24 September 2005, CM 2005 R:20.
Robinson, K.P., Stevick, P.T. and MacLeod, C.D. (2007). An Integrated Approach to Non-lethal Research on Minke Whales in European Waters. European Cetacean Society Spec. Public. Series 47: 8-13.
Rojano-Doñate, L., Mcdonald, B., Wisniewska, D., Johnson, M., Teilmann, J.,Wahlberg, M., Kristensen, J. and Madsen, P. (2018). High field metabolic rates of wild harbour porpoises. The Journal of Experimental Biology. 221.
Rolland, R. M., Parks, S. E., Hunt, K. E., Castellote, M., Corkeron, P. J., Nowacek, D. P., Wasser, S. K. and Kraus, S. D. (2012). Evidence that ship noise increases stress in right whales. Proceedings of the Royal Society B, 279 (1737), pp.2363-8. DOI:10.1098/rspb.2011.2429.
Rouse, S., Porter, J.S., Wilding, T.A. (2020). Artificial reef design affects benthic secondary productivity and provision of functional habitat. Ecol Evol. 10: 2122– 2130. https://doi.org/10.1002/ece3.6047. Accessed on: 19 March 2024.
Russell, D. J. F., Brasseur, S. M. J. M., Thompson, D., Hastie, G. D., Janik, V. M., Aarts, G., McClintock, B. T., Matthiopoulos, J., Moss, S. E. W. and McConnell, B. (2014). Marine mammals trace anthropogenic structures at sea. Current Biology, 24 (14), pp.R638-R639. DOI:10.1016/j.cub.2014.06.033.
Santos, M. B. and Pierce, G. J. (2003). The diet of harbour porpoise (Phocoena phocoena) in the northeast Atlantic: A review. Oceanography and Marine Biology, An Annual Review, Volume 41, pp.363-369.
Scheidat, M., Tougaard, J., Brasseur, S., Carstensen, J., van Polanen Petel, T., Teilmann, J. and Reijnders, P. (2011). Harbour porpoises (Phocoena phocoena) and wind farms: a case study in the Dutch North Sea. Environmental Research Letters, 6(2), p.025102.
Scott, B. (2022). Ecologically Sustainable Futures for Large-scale Renewables and How to Get There. International Marine Energy Journal, 5(1), pp.37-43.
Scottish Government (2024b). The Sandeel (Prohibition Of Fishing) (Scotland) Order 2024: business and regulatory impact assessment - final 2. Available at: https://www.gov.scot/publications/sandeel-prohibition-fishing-scotland-order-2024-island-communities-impact-assessment/. Accessed on: 27 March 2024.
Scottish Government, (2021). UK Dolphin and Porpoise Conservation Strategy: High Level report. Scottish Government, Edinburgh.
SEER (2022). Risk to marine life from marine debris and floating offshore wind cable systems. Report by National Renewable Energy Laboratory and Pacific Northwest National Laboratory for the U.S. Department of Energy, Wind Energy Technologies Office.
Skeate, E. R., Perrow, M. R. and Gilroy, J. J. (2012). Likely effects of construction of Scroby Sands offshore wind farm on a mixed population of harbour Phoca vitulina and grey Halichoerus grypus seals. Marine Pollution Bulletin, 64 (4), pp.872-881. DOI:10.1016/j.marpolbul.2012.01.029.
Sigray, P. and Andersson, M. (2011). Particle Motion Measured at an Operation Wind Turbine in Relation to Hearing Sensitivity in Fish. The Journal of the Acoustical Society of America. 130. 200-7.
Sparling, C.E. (2012). Seagreen Firth of Forth Round 3 Zone Marine Mammal Surveys. Report number SMRUL-ROY- 2012-006 to Royal Haskoning and Seagreen Wind Energy Ltd.
Special Committee on Seals (SCOS) (2018). Scientific Advice on Matters Related to the Management of Seal Populations: 2018. Available at SCOS-2018.pdf (st-andrews.ac.uk). Accessed 18 March 2024.
Statoil. (2015). Hywind Scotland Pilot Park: Environmental Statement (Full Report). Document Number A-100142-S35-EIAS-001. pp.462pp.
Stevens, A. (2023). Seal haul-out and telemetry data in relation to the Ossian Offshore Wind Farm. Document Number SMRUC-RPS-2023-011.
Teilmann, J., Tougaard, J. and Carstensen, J. (2006). Summary on harbour porpoise monitoring 1999-2006 around Nysted and Horns Rev Offshore Wind Farms. Report to Energi E2 A/S and Vattenfall A/S
Teilmann, J., Tougaard, J. and Carstensen, J. (2008). Effects from offshore wind farms on harbour porpoises in Denmark. Offshore wind farms and marine mammals: impacts & methodologies for assessing impacts, 50.
Tetley, M. J., Mitchelson-Jacob, E. G. and Robinson, K. P. (2008). The summer distribution of coastal minke whales (Balaenoptera acutorostrata) in the southern outer Moray Firth, NE Scotland, in relation to co-occurring mesoscale oceanographic features. Remote Sensing of Environment, 112(8), 3449–3454. https://doi.org/10.1016/J.RSE.2007.10.015.
Todd, G., Jodrey, A. and Stahlschmidt, Z. (2016). Immune activation influences the trade-off between thermoregulation and shelter use. Animal behaviour, 118, 27-32.
Tougaard, J., Carstensen, J., Damsgaard Henriksen, O. and Teilmann, J. (2003). Short-term effects of the construction of wind turbines on harbour porpoises at Horns Reef. Technical Report to Techwise A/S. Hedeselskabet, Roskilde
Verfuss, U. K., Sparling, C. E., Arnot, C., Judd, A. and Coyle, M. (2016). Review of Offshore Wind Farm Impact Monitoring and Mitigation with Regard to Marine Mammals. Springer New York.
Walsh, M. and Johnstone, A.D.F. (2006). Spawning behaviour and diel periodicity of egg production in captive Atlantic mackerel, Scomber scombrus L.*. Journal of Fish Biology, 40(6), pp. 939-50. Available at: DOI:10.1111/j.1095- 8649.1992.tb02639.x. Accessed on: 21 March 2024.
Walther, G-R. (2010). Community and ecosystem responses to recent climate change. Philosophical Transactions of the Royal Society Biology, 365(1549), pp.2019-2024.
Wanless, S., Harris, M. P., Newell, M. A., Speakman, J. R. and Daunt, F. (2018). Community-wide decline in the occurrence of lesser sandeels Ammodytes marinus in seabird chick diets at a North Sea colony. Marine Ecology Progress Series, 600. 193-206
Weir, C. R., Pollock, C., Cronin, C. and Taylor, S. (2001). Cetaceans of the Atlantic Frontier, north and west of Scotland. Continental Shelf Research, 21 (8), pp.1047-1071. DOI:https://doi.org/10.1016/S0278-4343(00)00124-2. Accessed on: 27 March 2024.
Weir, C., Stockin, K. and Pierce, G. (2007). Spatial and temporal trends in the distribution of harbour porpoises, white-beaked dolphins and minke whales off Aberdeenshire (UK), north-western North Sea. Journal of the Marine Biological Association of the United Kingdom. 87. 327 - 338. 10.1017/S0025315407052721.
Wilhelmsson, D., Malm, T., Thompson, R., Tchou, J., Sarantakos, G., McCormick, N., Luitjens, S., Gullström, M., Patterson Edwards, J.K., Amir, O. and Dubi, A. (2010). Greening Blue Energy: Identifying and Managing the Biodiversity Risks and Opportunities of Offshore Renewable Energy. Edited by Gland, Switzerland: IUCN. 102pp.
Winter H.V., Aarts G. and Van Keeken O.A. (2010) Residence time and behaviour of sole and cod in the Offshore Wind Farm Egmond aan Zee (OWEZ) IMARES, Wageningen YR Report number: C038/10, p 50.
Wright, P.J. and Bailey, M.C. (1996). Timing of hatching in Ammodytes marinus from Shetland waters and its significance to early growth and survivorship. Marine Biology, 126, 143-152.
Wright, P., Orpwood, J. and Scott, B. (2017). Impact of rising temperature on reproductive investment in a capital breeder: the lesser sandeel. Journal of Experimental Marine Biology and Ecology, 486, 52-58.
Wright, P.J., Pinnegar, J.K. and Fox, C. (2020) Impacts of climate change on fish, relevant to the coastal and marine environment around the UK. MCCIP Science Review 2020, 354–381.
[1] No specific major accidents and disasters receptors were identified and therefore to avoid duplication of receptors listed under other topic chapters, the chapter has not been included in this assessment.
[2] Inter-related effects specific to climatic effect receptors are discussed in a separate In Combination Climate Impacts (ICCI) document (volume 3, appendix 17.3).
[3] C = Construction, O = Operation and maintenance, D = Decommissioning.
[4] C = Construction, O = Operation and maintenance, D = Decommissioning.
[5] C = Construction, O = Operation and maintenance, D = Decommissioning.
[6] C = Construction, O = Operation and maintenance, D = Decommissioning
[7] C = Construction, O = Operation and maintenance, D = Decommissioning
[8] C = Construction, O = Operation and maintenance, D = Decommissioning
[9] C = Construction, O = Operation and maintenance, D = Decommissioning
[10] C = Construction, O = Operation and maintenance, D = Decommissioning
[11] C = Construction, O = Operation and maintenance, D = Decommissioning
[12] C = Construction, O = Operation and maintenance, D = Decommissioning
[13] C = Construction, O = Operation and maintenance, D = Decommissioning
[14] Plunge divers dive into the sea from a height to catch prey, whereas pursuit divers dive and can then swim underwater in pursuit of prey.